S100A4 promotes hepatocellular carcinogenesis by intensifying fibrosis-associated cancer cell stemness
- PMID: 32117590
- PMCID: PMC7028350
- DOI: 10.1080/2162402X.2020.1725355
S100A4 promotes hepatocellular carcinogenesis by intensifying fibrosis-associated cancer cell stemness
Abstract
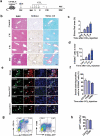
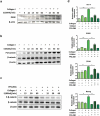
A cancer-promoting role of fibrogenesis in the liver has long been speculated; however, the molecular mechanisms regarding this phenomenon are largely unknown. We demonstrated in our previous study that macrophage-derived S100A4 promotes liver fibrosis via activation of hepatic stellate cells; however, whether and how S100A4 directly contributes to the development of fibrosis-associated liver cancer remains elusive. High expression of S100A4 in the fibrotic region was observed in human liver tumor tissues which associated with advanced disease severity. Through an established hepatocarcinogenesis model involving apparent liver fibrogenesis, we found that S100A4-deficient mice developed significantly less and smaller liver tumor nodules, with no change in the liver inflammation but decreased liver fibrosis and expression of stem cell markers in hepatocellular carcinoma (HCC) tissues. Mechanistically, S100A4 directly promoted stem cell-associated genes signatures in a way synergistic with its interacting protein, extracellular matrix component collagen I. This process is dependent on the receptor of advanced glycation end products (RAGE) and β-catenin signaling. Furthermore, the liver tumor sphere formation in vitro and tumor growth in vivo were greatly enhanced only when the cancer cells were pretreated with both S100A4 and collagen I. Our work firstly demonstrated a key role of S100A4 in synergy with extracellular matrix in the promotion of hepatocellular carcinoma by affecting the stemness of cancer cells.
Keywords: S100A4; collagen I; fibrosis; hepatocellular carcinogenesis; stemness.
© 2020 The Author(s). Published with license by Taylor & Francis Group, LLC.
Figures






References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
