A Phase I Trial of Intravesical Cabazitaxel, Gemcitabine and Cisplatin for the Treatment of Nonmuscle Invasive bacillus Calmette-Guérin Unresponsive or Recurrent/Relapsing Urothelial Carcinoma of the Bladder
- PMID: 32118506
- PMCID: PMC8464350
- DOI: 10.1097/JU.0000000000000919
A Phase I Trial of Intravesical Cabazitaxel, Gemcitabine and Cisplatin for the Treatment of Nonmuscle Invasive bacillus Calmette-Guérin Unresponsive or Recurrent/Relapsing Urothelial Carcinoma of the Bladder
Abstract
Purpose: For patients with bacillus Calmette-Guérin unresponsive or recurrent/relapsing nonmuscle invasive bladder cancer, multi-agent intravesical trials have been limited. In this study we investigate the safety of intravesical cabazitaxel, gemcitabine and cisplatin in the salvage setting.
Materials and methods: This was a dose escalation, drug escalation trial for patients with bacillus Calmette-Guérin unresponsive or recurrent/relapsing nonmuscle invasive bladder cancer who declined or were ineligible for radical cystectomy. All patients underwent a 6-week induction regimen of sequentially administered cabazitaxel, gemcitabine and cisplatin. Complete response was defined as no cancer on post-induction transurethral bladder tumor resection and negative urine cytology, while partial response allowed for positive cytology. Responders continued with maintenance cabazitaxel and gemcitabine monthly for the first year and bimonthly for the second year.
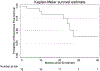
Results: A total of 18 patients were enrolled. Mean age was 71 years, median followup was 27.8 months (range 16.3 to 46.9) and mean number of previous rounds of intravesical therapies before trial enrollment was 3.7. Nine patients (50%) had received intravesical chemotherapy after bacillus Calmette-Guérin and 7 (39%) were previously treated in a phase I clinical trial setting. At enrollment 6 (33%) subjects had T1 disease and 13 (72%) had carcinoma in situ. There were no dose limiting toxicities. Initial partial and complete response rates were 94% and 89%, respectively. At 1 year recurrence-free survival was 0.83 (range 0.57 to 0.94) and at 2 years estimated recurrence-free survival was 0.64 (0.32 to 0.84).
Conclusions: In this high risk and highly pretreated cohort of bacillus Calmette-Guérin unresponsive or recurrent/relapsing nonmuscle invasive bladder cancer cases combination intravesical cabazitaxel, gemcitabine and cisplatin was a well tolerated and potentially effective regimen.
Keywords: administration; intravesical; urinary bladder neoplasms.
Figures
Comment in
-
Editorial Comment.J Urol. 2020 Aug;204(2):253. doi: 10.1097/JU.0000000000000919.01. Epub 2020 May 27. J Urol. 2020. PMID: 32459549 No abstract available.
References
-
- Ferlay J, Soerjomataram I, Dikshit R et al.: Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015; 136: E359. - PubMed
-
- National Cancer Institute: Cancer Stat Facts: Bladder Cancer. National Cancer Institute: Surveillance, Epidemiology, and End Results Program. Available at https://seer.cancer.gov/statfacts/html/urinb.html.
-
- Sylvester RJ, Brausi MA, Kirkels WJ et al.: Long-term efficacy results of EORTC genito-urinary group randomized phase 3 study 30911 comparing intravesical instillations of epirubicin, bacillus Calmette-Guerin, and bacillus Calmette-Guerin plus isoniazid in patients with intermediate- and high-risk stage Ta T1 urothelial carcinoma of the bladder. Eur Urol 2010; 57: 766. - PMC - PubMed
-
- Oddens J, Brausi M, Sylvester R et al.: Final results of an EORTC-GU cancers group randomized study of maintenance bacillus Calmette-Guerin in intermediate- and high-risk Ta, T1 papillary carcinoma of the urinary bladder: one-third dose versus full dose and 1 year versus 3 years of maintenance. Eur Urol 2013; 63: 462. - PubMed
-
- Lamm DL, Blumenstein BA, Crissman JD et al.: Maintenance bacillus Calmette-Guerin immunotherapy for recurrent Ta, T1 and carcinoma in situ transitional cell carcinoma of the bladder: a randomized Southwest Oncology Group study. J Urol 2000; 163: 1124. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical