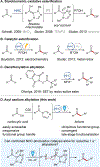
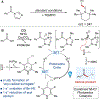
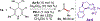Combined Photoredox and Carbene Catalysis for the Synthesis of Ketones from Carboxylic Acids
- PMID: 32119162
- PMCID: PMC7250732
- DOI: 10.1002/anie.202001824
Combined Photoredox and Carbene Catalysis for the Synthesis of Ketones from Carboxylic Acids
Abstract
As a key element in the construction of complex organic scaffolds, the formation of C-C bonds remains a challenge in the field of synthetic organic chemistry. Recent advancements in single-electron chemistry have enabled new methods for the formation of various C-C bonds. Disclosed herein is the development of a novel single-electron reduction of acyl azoliums for the formation of ketones from carboxylic acids. Facile construction of the acyl azolium in situ followed by a radical-radical coupling was made possible merging N-heterocyclic carbene (NHC) and photoredox catalysis. The utility of this protocol in synthesis was showcased in the late-stage functionalization of a variety of pharmaceutical compounds. Preliminary investigations using chiral NHCs demonstrate that enantioselectivity can be achieved, showcasing the advantages of this protocol over alternative methodologies.
Keywords: N-heterocyclic carbenes; ketones; photochemistry; radicals; reaction mechanisms.
© 2020 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures
Similar articles
-
Light-Driven Carbene Catalysis for the Synthesis of Aliphatic and α-Amino Ketones.Angew Chem Int Ed Engl. 2021 Aug 9;60(33):17925-17931. doi: 10.1002/anie.202105354. Epub 2021 Jul 14. Angew Chem Int Ed Engl. 2021. PMID: 34097802 Free PMC article.
-
Bio- and Medicinally Compatible α-Amino-Acid Modification via Merging Photoredox and N-Heterocyclic Carbene Catalysis.Org Lett. 2020 Aug 21;22(16):6370-6375. doi: 10.1021/acs.orglett.0c02202. Epub 2020 Aug 5. Org Lett. 2020. PMID: 32806184
-
Oxidative γ-addition of enals to trifluoromethyl ketones: enantioselectivity control via Lewis acid/N-heterocyclic carbene cooperative catalysis.J Am Chem Soc. 2012 May 30;134(21):8810-3. doi: 10.1021/ja303618z. Epub 2012 May 17. J Am Chem Soc. 2012. PMID: 22571795
-
Bifunctional N-Heterocyclic Carbenes Derived from l-Pyroglutamic Acid and Their Applications in Enantioselective Organocatalysis.Acc Chem Res. 2020 Mar 17;53(3):690-702. doi: 10.1021/acs.accounts.9b00635. Epub 2020 Mar 6. Acc Chem Res. 2020. PMID: 32142245 Review.
-
A continuum of progress: applications of N-hetereocyclic carbene catalysis in total synthesis.Angew Chem Int Ed Engl. 2012 Nov 19;51(47):11686-98. doi: 10.1002/anie.201203704. Epub 2012 Oct 16. Angew Chem Int Ed Engl. 2012. PMID: 23074146 Free PMC article. Review.
Cited by
-
Combined Photoredox and Carbene Catalysis for the Synthesis of γ-Aryloxy Ketones.Adv Synth Catal. 2022 Feb 1;364(3):518-524. doi: 10.1002/adsc.202101354. Epub 2021 Dec 1. Adv Synth Catal. 2022. PMID: 35431717 Free PMC article.
-
N-Heterocyclic carbene catalytic 1,2-boron migrative acylation accelerated by photocatalysis.Sci Adv. 2024 Jul 26;10(30):eadn8401. doi: 10.1126/sciadv.adn8401. Epub 2024 Jul 24. Sci Adv. 2024. PMID: 39047096 Free PMC article.
-
Photoredox cooperative N-heterocyclic carbene/palladium-catalysed alkylacylation of alkenes.Nat Commun. 2022 Sep 30;13(1):5754. doi: 10.1038/s41467-022-33444-0. Nat Commun. 2022. PMID: 36180483 Free PMC article.
-
Suzuki-type cross-coupling of alkyl trifluoroborates with acid fluoride enabled by NHC/photoredox dual catalysis.Chem Sci. 2022 Jan 21;13(9):2584-2590. doi: 10.1039/d1sc06102j. eCollection 2022 Mar 2. Chem Sci. 2022. PMID: 35356672 Free PMC article.
-
Synthesis of Cyclohexanones by a Tandem Photocatalyzed Annulation.J Am Chem Soc. 2022 Apr 27;144(16):7030-7037. doi: 10.1021/jacs.1c13105. Epub 2022 Mar 22. J Am Chem Soc. 2022. PMID: 35316053 Free PMC article.
References
-
- Breslow R, J. Am. Chem. Soc 1958, 80, 3719–3726;
- Maki BE, Chan A, Phillips EM, Scheidt KA, Org. Lett 2007, 9, 371–374; - PubMed
- Ukai T, Tanaka R, Dokawa T, J. Pharm. Soc. Jpn 1943, 63, 296–300.
-
- Guin J, De Sarkar S, Grimme S, Studer A, Angew. Chem. Int. Ed 2008, 47, 8727–8730. - PubMed
-
- De Sarkar S, Grimme S, Studer A, J. Am. Chem. Soc 2010, 132, 1190–1191. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials