Enhancing the Thermo-Stability and Anti-Bacterium Activity of Lysozyme by Immobilization on Chitosan Nanoparticles
- PMID: 32121010
- PMCID: PMC7084273
- DOI: 10.3390/ijms21051635
Enhancing the Thermo-Stability and Anti-Bacterium Activity of Lysozyme by Immobilization on Chitosan Nanoparticles
Abstract
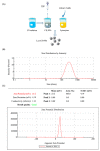
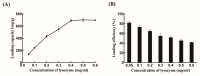
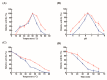
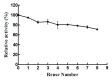
The recent emergence of antibiotic-resistant bacteria requires the development of new antibiotics or new agents capable of enhancing antibiotic activity. Lysozyme degrades bacterial cell wall without involving antibiotic resistance and has become a new antibacterial strategy. However, direct use of native, active proteins in clinical settings is not practical as it is fragile under various conditions. In this study, lysozyme was integrated into chitosan nanoparticles (CS-NPs) by the ionic gelation technique to obtain lysozyme immobilized chitosan nanoparticles (Lys-CS-NPs) and then characterized by dynamic light scattering (DLS) and transmission electron microscopy (TEM), which showed a small particle size (243.1 ± 2.1 nm) and positive zeta potential (22.8 ± 0.2 mV). The immobilization significantly enhanced the thermal stability and reusability of lysozyme. In addition, compared with free lysozyme, Lys-CS-NPs exhibited superb antibacterial properties according to the results of killing kinetics in vitro and measurement of the minimum inhibitory concentration (MIC) of CS-NPs and Lys-CS-NPs against Pseudomonas aeruginosa (P. aeruginosa), Klebsiella pneumoniae (K. pneumoniae), Escherichia coli (E. coli), and Staphylococcus aureus (S. aureus). These results suggest that the integration of lysozyme into CS-NPs will create opportunities for the further potential applications of lysozyme as an anti-bacterium agent.
Keywords: anti-bacterium activity; chitosan nanoparticles; immobilization; lysozyme.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Integration of lysozyme into chitosan nanoparticles for improving antibacterial activity.Carbohydr Polym. 2017 Jan 2;155:192-200. doi: 10.1016/j.carbpol.2016.08.076. Epub 2016 Aug 25. Carbohydr Polym. 2017. PMID: 27702504
-
Incorporation of lysozyme into cellulose nanocrystals stabilized β-chitosan nanoparticles with enhanced antibacterial activity.Carbohydr Polym. 2020 May 15;236:115974. doi: 10.1016/j.carbpol.2020.115974. Epub 2020 Feb 11. Carbohydr Polym. 2020. PMID: 32172828
-
Enhancing the Thermo-Stability and Anti-Biofilm Activity of Alginate Lyase by Immobilization on Low Molecular Weight Chitosan Nanoparticles.Int J Mol Sci. 2019 Sep 14;20(18):4565. doi: 10.3390/ijms20184565. Int J Mol Sci. 2019. PMID: 31540110 Free PMC article.
-
Research and Application of Chitosan Nanoparticles in Orthopedic Infections.Int J Nanomedicine. 2024 Jul 2;19:6589-6602. doi: 10.2147/IJN.S468848. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38979535 Free PMC article. Review.
-
Chitosan-based nanoparticles against bacterial infections.Carbohydr Polym. 2021 Jan 1;251:117108. doi: 10.1016/j.carbpol.2020.117108. Epub 2020 Sep 20. Carbohydr Polym. 2021. PMID: 33142645 Review.
Cited by
-
Bacteriophage Endolysin: A Powerful Weapon to Control Bacterial Biofilms.Protein J. 2023 Oct;42(5):463-476. doi: 10.1007/s10930-023-10139-z. Epub 2023 Jul 25. Protein J. 2023. PMID: 37490161 Review.
-
Integration of Lysin into Chitosan Nanoparticles for Improving Bacterial Biofilm Inhibition.Appl Biochem Biotechnol. 2024 Mar;196(3):1592-1611. doi: 10.1007/s12010-023-04627-2. Epub 2023 Jul 12. Appl Biochem Biotechnol. 2024. PMID: 37436548
-
Lactobacillus rhamnosus and Staphylococcus epidermidis in gut microbiota: in vitro antimicrobial resistance.AMB Express. 2022 Oct 3;12(1):128. doi: 10.1186/s13568-022-01468-w. AMB Express. 2022. PMID: 36190582 Free PMC article.
-
Expression of BSN314 lysozyme genes in Escherichia coli BL21: a study to demonstrate microbicidal and disintegarting potential of the cloned lysozyme.Braz J Microbiol. 2024 Mar;55(1):215-233. doi: 10.1007/s42770-023-01219-4. Epub 2023 Dec 26. Braz J Microbiol. 2024. PMID: 38146050 Free PMC article.
-
New advances in biological preservation technology for aquatic products.NPJ Sci Food. 2025 Feb 3;9(1):15. doi: 10.1038/s41538-025-00372-4. NPJ Sci Food. 2025. PMID: 39900935 Free PMC article. Review.
References
-
- Wen S., Yao D., Liu X., Wang F. A novel fluorescence resonance energy transfer-based high-throughput screening method for generation of lysozyme with improved antimicrobial activity against Escherichia coli strains. J. Agric. Food. Chem. 2019;67:12584–12589. doi: 10.1021/acs.jafc.9b05364. - DOI - PubMed
MeSH terms
Substances
Grants and funding
- 31900031/National Natural Science Foundation of China (NSFC)
- ZR2019BD027/Shandong Provincial Natural Science Foundation
- 2019H1D3A1A01102881/Korea Research Fellowship Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT
- MBSMAT-2019-02/Key Lab of Marine Bioactive Substance and Modern Analytical Technique (SOA)
LinkOut - more resources
Full Text Sources
Medical

