Do children and adolescents with completely resected alveolar rhabdomyosarcoma require adjuvant radiation? A report from the Children's Oncology Group
- PMID: 32124549
- PMCID: PMC7325864
- DOI: 10.1002/pbc.28243
Do children and adolescents with completely resected alveolar rhabdomyosarcoma require adjuvant radiation? A report from the Children's Oncology Group
Abstract
Background: The role of adjuvant radiotherapy (RT) remains unclear in patients with localized, completely resected (group I) alveolar rhabdomyosarcoma (ARMS).
Procedure: Patients with group I ARMS enrolled on any one of three prior Children's Oncology Group (COG) clinical trials (D9602, D9803, or ARST0531) were analyzed. All patients received systemic chemotherapy and 36 Gy adjuvant RT (if given) to the primary site at week 12 or week 4 for D9602/D9803 and ARST0531, respectively.
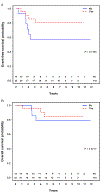
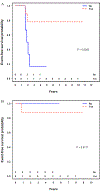
Results: Thirty-six patients with group I ARMS were treated on D9602 (n = 6), D9803 (n = 17), or ARST0531 (n = 13), of whom 24 (67%) were male. The median age was 4.1 years (range, 0.8-45.8). Twenty (56%) patients had an unfavorable primary site, and 10 (28%) had tumors > 5 cm. FOXO1-fusion status was negative, positive, and unknown in 10 (28%), 15 (42%), and 11 (30%) tumors, respectively. Twenty-two (61%) patients received RT. Overall, the four-year event-free survival (EFS) and overall survival (OS) were 70.8% and 88.3%, respectively. Patients with FOXO1 positivity who received RT had superior EFS compared with those who did not (77.8% vs 16.7%; P = 0.03). Among 10 patients who were FOXO1 negative, the outcome was similar with or without RT.
Conclusions: Although limited by a small sample size, data from this study support the routine use of adjuvant RT in patients with FOXO1-positive disease even after complete resection. Additionally, omitting adjuvant RT is rational for patients with FOXO1-negative ARMS and will be prospectively investigated in the current COG trial ARST1431.
Keywords: FOXO1; radiation; rhabdomyosarcoma.
© 2020 Wiley Periodicals, Inc.
Conflict of interest statement
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
Figures
Similar articles
-
Alveolar rhabdomyosarcoma with regional nodal involvement: Results of a combined analysis from two cooperative groups.Pediatr Blood Cancer. 2021 Mar;68(3):e28832. doi: 10.1002/pbc.28832. Epub 2020 Nov 27. Pediatr Blood Cancer. 2021. PMID: 33245207 Free PMC article.
-
Increased local failure for patients with intermediate-risk rhabdomyosarcoma on ARST0531: A report from the Children's Oncology Group.Cancer. 2019 Sep 15;125(18):3242-3248. doi: 10.1002/cncr.32204. Epub 2019 Jun 7. Cancer. 2019. PMID: 31174239 Free PMC article.
-
PAX-FOXO1 fusion status drives unfavorable outcome for children with rhabdomyosarcoma: a children's oncology group report.Pediatr Blood Cancer. 2013 Sep;60(9):1411-7. doi: 10.1002/pbc.24532. Epub 2013 Mar 22. Pediatr Blood Cancer. 2013. PMID: 23526739 Free PMC article. Clinical Trial.
-
Volumetric considerations in radiotherapy for pediatric parameningeal rhabdomyosarcomas.Int J Radiat Oncol Biol Phys. 2003 Apr 1;55(5):1294-9. doi: 10.1016/s0360-3016(02)04290-6. Int J Radiat Oncol Biol Phys. 2003. PMID: 12654440 Review.
-
Clinical group and modified TNM stage for rhabdomyosarcoma: A review from the Children's Oncology Group.Pediatr Blood Cancer. 2022 Jun;69(6):e29644. doi: 10.1002/pbc.29644. Epub 2022 Mar 6. Pediatr Blood Cancer. 2022. PMID: 35253352 Free PMC article. Review.
Cited by
-
Clinical features and prognosis of paediatric rhabdomyosarcoma with bone marrow metastasis: a single Centre experiences in China.BMC Pediatr. 2021 Oct 21;21(1):463. doi: 10.1186/s12887-021-02904-9. BMC Pediatr. 2021. PMID: 34670517 Free PMC article.
References
-
- Perez EA, Kassira N, Cheung MC, et al. Rhabdomyosarcoma in children: a SEER population based study. J Surg Res. 2011;170(2):e243–e251. - PubMed
-
- Raney RB, Anderson JR, Barr FG, et al. Rhabdomyosarcoma and undifferentiated sarcoma in the first two decades of life: a selective review of intergroup rhabdomyosarcoma study group experience and rationale for Intergroup Rhabdomyosarcoma Study V. J Pediatr Hematol Oncol. 2001;23(4):215–220. - PubMed
-
- Maurer HM, Beltangady M, Gehan EA, et al. The Intergroup Rhabdomyosarcoma Study I. A final report. Cancer. 1988;61(2):209–220. - PubMed
-
- Maurer HM, Gehan EA, Beltangady M, et al. The Intergroup Rhabdomyosarcoma Study II. Cancer. 1993;71(5):1904–1922. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous