Evaluation of Germline Genetic Testing Criteria in a Hospital-Based Series of Women With Breast Cancer
- PMID: 32125938
- PMCID: PMC7193748
- DOI: 10.1200/JCO.19.02190
Evaluation of Germline Genetic Testing Criteria in a Hospital-Based Series of Women With Breast Cancer
Abstract
Purpose: To determine the sensitivity and specificity of genetic testing criteria for the detection of germline pathogenic variants in women with breast cancer.
Materials and methods: Women with breast cancer enrolled in a breast cancer registry at a tertiary cancer center between 2000 and 2016 were evaluated for germline pathogenic variants in 9 breast cancer predisposition genes (ATM, BRCA1, BRCA2, CDH1, CHEK2, NF1, PALB2, PTEN, and TP53). The performance of the National Comprehensive Cancer Network (NCCN) hereditary cancer testing criteria was evaluated relative to testing of all women as recommended by the American Society of Breast Surgeons.
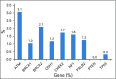
Results: Of 3,907 women, 1,872 (47.9%) meeting NCCN criteria were more likely to carry a pathogenic variant in 9 predisposition genes compared with women not meeting criteria (9.0% v 3.5%; P < .001). Of those not meeting criteria (n = 2,035), 14 (0.7%) had pathogenic variants in BRCA1 or BRCA2. The sensitivity of NCCN criteria was 70% for 9 predisposition genes and 87% for BRCA1 and BRCA2, with a specificity of 53%. Expansion of the NCCN criteria to include all women diagnosed with breast cancer at ≤ 65 years of age achieved > 90% sensitivity for the 9 predisposition genes and > 98% sensitivity for BRCA1 and BRCA2.
Conclusion: A substantial proportion of women with breast cancer carrying germline pathogenic variants in predisposition genes do not qualify for testing by NCCN criteria. Expansion of NCCN criteria to include all women diagnosed at ≤ 65 years of age improves the sensitivity of the selection criteria without requiring testing of all women with breast cancer.
Figures


References
-
- NCCN Clinical Practice Guidelines in Oncology . Genetic/familial high-risk assessment: Breast, ovarian, and pancreatic Version 1. National Comprehensive Cancer Network: ; https://www.nccn.org/professionals/physician_gls/pdf/genetics_bop.pdf - PubMed
-
- Yang S, Axilbund JE, O’Leary E, et al. Underdiagnosis of hereditary breast and ovarian cancer in Medicare patients: Genetic testing criteria miss the mark. Ann Surg Oncol. 2018;25:2925–2931. - PubMed
-
- doi: 10.1038/s41436-019-0712-x. Pal T, Agnese D, Daly M, et al: Points to consider: Is there evidence to support BRCA1/2 and other inherited breast cancer genetic testing for all breast cancer patients? A statement of the American College of Medical Genetics and Genomics (ACMG). Genet Med . [Epub ahead of print on December 13, 2019] - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

