Inactivation of Plasmodium falciparum in whole blood using the amustaline and glutathione pathogen reduction technology
- PMID: 32129497
- PMCID: PMC7187285
- DOI: 10.1111/trf.15734
Inactivation of Plasmodium falciparum in whole blood using the amustaline and glutathione pathogen reduction technology
Abstract
Background: Risk of transfusion-transmitted (TT) malaria is mainly associated with whole blood (WB) or red blood cell (RBC) transfusion. Risk mitigation relies mostly on donor deferral while a limited number of countries perform blood testing, both negatively impacting blood availability. This study investigated the efficacy of the pathogen reduction system using amustaline and glutathione (GSH) to inactivate Plasmodium falciparum in WB.
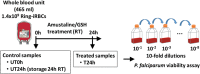
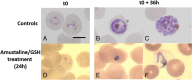
Study design and methods: WB units were spiked with ring stage P. falciparum infected RBCs. Parasite loads were measured in samples at time of infection, after 24 hours at room temperature (RT), and after a 24-hour incubation at RT post-treatment with 0.2 mM amustaline and 2 mM GSH. Serial 10-fold dilutions of the samples were inoculated to RBC cultures and maintained up to 4 weeks. Parasitemia was quantified by cytometry.
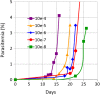
Results: The P. falciparum viability assay has a limit of detection of a single live parasite per sample. Input parasite titer was >5.7 log10 TCID50 per mL. A 24-hour incubation at RT paused parasite development in controls, but they retained viability and infectivity when tested in culture. In contrast, no infectious parasites were detected in the amustaline/GSH-treated sample after 4 weeks of culture.
Conclusion: A robust level of P. falciparum inactivation was achieved in WB using amustaline/GSH treatment. Parasite log reduction was >5.7 log10 TCID50 per mL. Development of such a pathogen reduction system may provide an opportunity to reduce the risk of TT malaria and improve blood availability.
© 2020 The Authors. Transfusion published by Wiley Periodicals, Inc. on behalf of AABB.
Conflict of interest statement
MCL and YAG are employees of Cerus Corporation; AL and AS were employees of Cerus Corporation at the time this study was executed. CS, PG, and SAED declare no conflict of interest relevant to this manuscript.
Figures
Similar articles
-
Robust inactivation of Plasmodium falciparum in red blood cell concentrates using amustaline and glutathione pathogen reduction.Transfusion. 2022 May;62(5):1073-1083. doi: 10.1111/trf.16867. Epub 2022 Apr 6. Transfusion. 2022. PMID: 35385146 Free PMC article.
-
Red blood cells treated with the amustaline (S-303) pathogen reduction system: a transfusion study in cardiac surgery.Transfusion. 2018 Apr;58(4):905-916. doi: 10.1111/trf.14528. Epub 2018 Mar 1. Transfusion. 2018. PMID: 29498049 Clinical Trial.
-
Pathogen inactivation of Dengue virus in red blood cells using amustaline and glutathione.Transfusion. 2017 Dec;57(12):2888-2896. doi: 10.1111/trf.14318. Epub 2017 Sep 16. Transfusion. 2017. PMID: 28921542
-
Towards pathogen inactivation of red blood cells and whole blood targeting viral DNA/RNA: design, technologies, and future prospects for developing countries.Blood Transfus. 2017 Oct;15(6):512-521. doi: 10.2450/2017.0344-16. Epub 2017 Apr 13. Blood Transfus. 2017. PMID: 28488960 Free PMC article. Review.
-
The pathogenesis of Plasmodium falciparum malaria in humans: insights from splenic physiology.Blood. 2011 Jan 13;117(2):381-92. doi: 10.1182/blood-2010-04-202911. Epub 2010 Sep 17. Blood. 2011. PMID: 20852127 Free PMC article. Review.
Cited by
-
Robust inactivation of Plasmodium falciparum in red blood cell concentrates using amustaline and glutathione pathogen reduction.Transfusion. 2022 May;62(5):1073-1083. doi: 10.1111/trf.16867. Epub 2022 Apr 6. Transfusion. 2022. PMID: 35385146 Free PMC article.
-
Impact of different pathogen reduction technologies on the biochemistry, function, and clinical effectiveness of platelet concentrates: An updated view during a pandemic.Transfusion. 2022 Jan;62(1):227-246. doi: 10.1111/trf.16747. Epub 2021 Dec 6. Transfusion. 2022. PMID: 34870335 Free PMC article. Review.
-
Transfusion-Transmitted Malaria and Mitigation Strategies in Nonendemic Regions.Transfus Med Hemother. 2022 Jul 15;49(4):205-217. doi: 10.1159/000525414. eCollection 2022 Aug. Transfus Med Hemother. 2022. PMID: 36159954 Free PMC article. Review.
-
Inactivation of SARS-CoV-2 in All Blood Components Using Amotosalen/Ultraviolet A Light and Amustaline/Glutathione Pathogen Reduction Technologies.Pathogens. 2022 Apr 28;11(5):521. doi: 10.3390/pathogens11050521. Pathogens. 2022. PMID: 35631042 Free PMC article.
-
Malaria : Stellungnahmen des Arbeitskreises Blut des Bundesministeriums für Gesundheit.Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz. 2022 Apr;65(4):512-524. doi: 10.1007/s00103-022-03490-6. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz. 2022. PMID: 35119479 Free PMC article. German. No abstract available.
References
-
- WHO. World malaria report 2019 [Internet]. 2019 [cited 2020 January 16]. Available from: https://apps.who.int/iris/handle/10665/330011
-
- European Centre for Disease Prevention and Control. Malaria – Annual Epidemiological Report for 2017 [cited 2020 January 16]. Annu Epidemiol Rep Commun Dis Eur [Internet]. 2019;393(July):1–6. Available from: https://www.ecdc.europa.eu/en/publications-data/malaria-annual-epidemiol...
-
- Kitchen AD, Chiodini PL. Malaria and blood transfusion. Vox Sang 2006;90:77‐84. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

