Disentangling group specific QTL allele effects from genetic background epistasis using admixed individuals in GWAS: An application to maize flowering
- PMID: 32130208
- PMCID: PMC7075643
- DOI: 10.1371/journal.pgen.1008241
Disentangling group specific QTL allele effects from genetic background epistasis using admixed individuals in GWAS: An application to maize flowering
Abstract
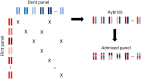
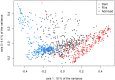
When handling a structured population in association mapping, group-specific allele effects may be observed at quantitative trait loci (QTLs) for several reasons: (i) a different linkage disequilibrium (LD) between SNPs and QTLs across groups, (ii) group-specific genetic mutations in QTL regions, and/or (iii) epistatic interactions between QTLs and other loci that have differentiated allele frequencies between groups. We present here a new genome-wide association (GWAS) approach to identify QTLs exhibiting such group-specific allele effects. We developed genetic materials including admixed progeny from different genetic groups with known genome-wide ancestries (local admixture). A dedicated statistical methodology was developed to analyze pure and admixed individuals jointly, allowing one to disentangle the factors causing the heterogeneity of allele effects across groups. This approach was applied to maize by developing an inbred "Flint-Dent" panel including admixed individuals that was evaluated for flowering time. Several associations were detected revealing a wide range of configurations of allele effects, both at known flowering QTLs (Vgt1, Vgt2 and Vgt3) and new loci. We found several QTLs whose effect depended on the group ancestry of alleles while others interacted with the genetic background. Our GWAS approach provides useful information on the stability of QTL effects across genetic groups and can be applied to a wide range of species.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Adaptation of maize to temperate climates: mid-density genome-wide association genetics and diversity patterns reveal key genomic regions, with a major contribution of the Vgt2 (ZCN8) locus.PLoS One. 2013 Aug 30;8(8):e71377. doi: 10.1371/journal.pone.0071377. eCollection 2013. PLoS One. 2013. PMID: 24023610 Free PMC article.
-
Identifying QTLs involved in hybrid performance and heterotic group complementarity: new GWAS models applied to factorial and admixed diallel maize hybrid panels.Theor Appl Genet. 2023 Oct 10;136(11):219. doi: 10.1007/s00122-023-04431-w. Theor Appl Genet. 2023. PMID: 37816986 Free PMC article.
-
Association mapping for cold tolerance in two large maize inbred panels.BMC Plant Biol. 2016 Jun 6;16(1):127. doi: 10.1186/s12870-016-0816-2. BMC Plant Biol. 2016. PMID: 27267760 Free PMC article.
-
Entering the second century of maize quantitative genetics.Heredity (Edinb). 2014 Jan;112(1):30-8. doi: 10.1038/hdy.2013.6. Epub 2013 Mar 6. Heredity (Edinb). 2014. PMID: 23462502 Free PMC article. Review.
-
Detecting epistasis in human complex traits.Nat Rev Genet. 2014 Nov;15(11):722-33. doi: 10.1038/nrg3747. Epub 2014 Sep 9. Nat Rev Genet. 2014. PMID: 25200660 Review.
Cited by
-
Understanding interactions between risk factors, and assessing the utility of the additive and multiplicative models through simulations.PLoS One. 2021 Apr 26;16(4):e0250282. doi: 10.1371/journal.pone.0250282. eCollection 2021. PLoS One. 2021. PMID: 33901204 Free PMC article.
-
Perspectives on Applications of Hierarchical Gene-To-Phenotype (G2P) Maps to Capture Non-stationary Effects of Alleles in Genomic Prediction.Front Plant Sci. 2021 Jun 4;12:663565. doi: 10.3389/fpls.2021.663565. eCollection 2021. Front Plant Sci. 2021. PMID: 34149761 Free PMC article.
-
Improvement in genomic prediction of maize with prior gene ontology information depends on traits and environmental conditions.Plant Genome. 2025 Mar;18(1):e20553. doi: 10.1002/tpg2.20553. Plant Genome. 2025. PMID: 39779652 Free PMC article.
-
Evaluating the Genetic Background Effect on Dissecting the Genetic Basis of Kernel Traits in Reciprocal Maize Introgression Lines.Genes (Basel). 2023 May 6;14(5):1044. doi: 10.3390/genes14051044. Genes (Basel). 2023. PMID: 37239404 Free PMC article.
-
Modeling allelic diversity of multiparent mapping populations affects detection of quantitative trait loci.G3 (Bethesda). 2022 Mar 4;12(3):jkac011. doi: 10.1093/g3journal/jkac011. G3 (Bethesda). 2022. PMID: 35100382 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

