Sensitive determination of malondialdehyde in rat prostate by high performance liquid chromatography with fluorescence detection
- PMID: 32132642
- PMCID: PMC7055245
- DOI: 10.1038/s41598-020-61074-3
Sensitive determination of malondialdehyde in rat prostate by high performance liquid chromatography with fluorescence detection
Abstract
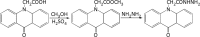
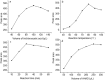
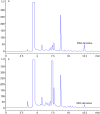
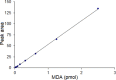
An excellent pre-column fluorescent derivatization reagent N-acetylhydrazine acridone for the quantitative determination of malondialdehyde was synthesized. Malondialdehyde was derivatized at 80 °C for 30 min in the presence of trichloroacetic acid. The separation of the derivative was performed on an Agilent ZORBAX SB-C18 column in conjunction with gradient elution. The excitation and emission wavelengths were 370 nm and 420 nm, respectively. The developed method demonstrated good linear relationship in the range of 0.02 pmol to 2.5 pmol (r = 0.9998). The calculated limit of detection and limit of quantification were 2.5 fmol and 8.3 fmol, respectively. The analytical precisions of the method were in the range of 1.36-2.27% (intra-day) and 2.36-3.92% (inter-day) respectively. The method was sensitive, specific and simple. It was successfully implemented to analysis the malondialdehyde in rat prostate.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Rezaei Z, Jamshidzadeh A, Sanati E. A rapid and sensitive method for the determination of malondialdehyde as its hydralazine derivative in human plasma using high performance liquid chromatography. Anal. Methods. 2013;12(5):2995–2999. doi: 10.1039/C3AY26525K. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources