Immunization against Leishmania major infection in BALB/c mice using a subunit-based DNA vaccine derived from TSA, LmSTI1, KMP11, and LACK predominant antigens
- PMID: 32133069
- PMCID: PMC7043880
- DOI: 10.22038/IJBMS.2019.14051
Immunization against Leishmania major infection in BALB/c mice using a subunit-based DNA vaccine derived from TSA, LmSTI1, KMP11, and LACK predominant antigens
Abstract
Objectives: To design a multivalent DNA vaccine encoding the most immunogenic regions of the Leishmania major antigens including TSA (Thiol-specific antioxidant protein), LmSTI1 (Leishmania major stress-inducible protein1), LACK (Leishmania homologue of receptors for activated C Kinase), and KMP11 (kinetoplastid membrane protein-11) on BALB/c mice.
Materials and methods: The chimeric construct was generated including the most immunogenic epitopes containing a combination of domains and oligopeptides of the aforementioned genes. The construct was cloned into pcDNA 3.1 plasmid and named "pleish-dom." Following intramuscular injection of mice, the capability of the vector pleish-dom alone and with pIL-12 (expressing murine IL-12) to raise protective cytokines and parasite burden was evaluated in the BALB/c mice as a susceptible animal model against L. major.
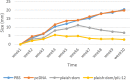
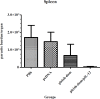
Results: The immunized mice with pleish-dom/pIL-12 showed the highest and the lowest levels of interferon-gamma (IFN-γ) and interleukin-10 (IL-10), as well as the lowest leishmanin skin test (LST) reactions, were found through 8 weeks post-infection.
Conclusion: Although the obtained DNA vaccine from the immunogenic chimeric protein of L. major antigens was able to induce a high level of IFN-γ, it partially protected mice against L. major. However co-administration with pIL-12 led to shift immune response to Th1 phenotype, granuloma formation, and lowered parasite burden, and consequently distinct protection was found.
Keywords: BALB/c mice; Cytokines; DNA; Epitopes; Leishmania major; Vaccines.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures



Similar articles
-
Vaccination with plasmid DNA encoding TSA/LmSTI1 leishmanial fusion proteins confers protection against Leishmania major infection in susceptible BALB/c mice.Infect Immun. 2002 Jun;70(6):2828-36. doi: 10.1128/IAI.70.6.2828-2836.2002. Infect Immun. 2002. PMID: 12010969 Free PMC article.
-
Evaluation of a new live recombinant vaccine against cutaneous leishmaniasis in BALB/c mice.Parasit Vectors. 2020 Aug 12;13(1):415. doi: 10.1186/s13071-020-04289-7. Parasit Vectors. 2020. PMID: 32787908 Free PMC article.
-
Immunization with a polyprotein vaccine consisting of the T-Cell antigens thiol-specific antioxidant, Leishmania major stress-inducible protein 1, and Leishmania elongation initiation factor protects against leishmaniasis.Infect Immun. 2002 Aug;70(8):4215-25. doi: 10.1128/IAI.70.8.4215-4225.2002. Infect Immun. 2002. PMID: 12117930 Free PMC article.
-
DNA vaccination with a plasmid encoding LACK-TSA fusion against Leishmania major infection in BALB/c mice.Malays J Pathol. 2017 Dec;39(3):267-275. Malays J Pathol. 2017. PMID: 29279589
-
A non-pathogenic Leishmania tarentolae vector based- HCV polytope DNA vaccine elicits potent and long lasting Th1 and CTL responses in BALB/c mice model.Mol Immunol. 2019 Jul;111:152-161. doi: 10.1016/j.molimm.2019.04.009. Epub 2019 May 1. Mol Immunol. 2019. PMID: 31054409
Cited by
-
Immunomodulatory activity of Trypanosoma cruzi recombinant antigen combination TSA-1-C4 and Tc24-C4 induce activation of macrophages and CD8+ T cells.Parasitol Res. 2025 Jan 24;124(1):12. doi: 10.1007/s00436-025-08453-9. Parasitol Res. 2025. PMID: 39853538 Free PMC article.
-
A second generation leishmanization vaccine with a markerless attenuated Leishmania major strain using CRISPR gene editing.Nat Commun. 2020 Jul 10;11(1):3461. doi: 10.1038/s41467-020-17154-z. Nat Commun. 2020. PMID: 32651371 Free PMC article.
-
Exploring the out of sight antigens of SARS-CoV-2 to design a candidate multi-epitope vaccine by utilizing immunoinformatics approaches.Vaccine. 2020 Nov 10;38(48):7612-7628. doi: 10.1016/j.vaccine.2020.10.016. Epub 2020 Oct 9. Vaccine. 2020. PMID: 33082015 Free PMC article.
-
Amphotericin B-Loaded Extracellular Vesicles Derived from Leishmania major Enhancing Cutaneous Leishmaniasis Treatment through In Vitro and In Vivo Studies.Iran J Parasitol. 2023 Oct-Dec;18(4):514-525. doi: 10.18502/ijpa.v18i4.14260. Iran J Parasitol. 2023. PMID: 38169565 Free PMC article.
-
Bioinformatics analysis for the purpose of designing a novel multi-epitope DNA vaccine against Leishmania major.Sci Rep. 2022 Oct 27;12(1):18119. doi: 10.1038/s41598-022-22646-7. Sci Rep. 2022. PMID: 36302830 Free PMC article.
References
-
- O mondiale de la Santé, World Health Organization. Leishmaniasis in high-burden countries: an epidemiological update based on data reported in 2014. Wkly Epidemiol Rec. 2016;91:287–296. - PubMed
-
- Leishmaniases WECotCot, Meeting WECotCotL, World Health Organization. Control of the Leishmaniases: Report of a WHO Expert Committee. World Health Organization . 1990 - PubMed
-
- Khanjani N, González U, Leonardi-Bee J, Mohebali M, Saffari M, Khamesipour A. Vaccines for preventing cutaneous leishmaniasis. Cochrane Database Syst Rev . 2009
-
- Nadim A, Javadian E, Mohebali M. The experience of leishmanization in the Islamic Republic of Iran. East Mediterr Health J. 1997;3:284–290.
-
- Mohebali M, Khamesipour A, Mobedi I, Zarei Z, Hashemi-Fesharki R. Double-blind randomized efficacy field trial of alum precipitated autoclaved Leishmania major vaccine mixed with BCG against canine visceral leishmaniasis in Meshkin-Shahr district, IR Iran. Vaccine. 2004;22:4097–4100. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
