Disruption of Intracellular Calcium Homeostasis as a Therapeutic Target Against Trypanosoma cruzi
- PMID: 32133302
- PMCID: PMC7040492
- DOI: 10.3389/fcimb.2020.00046
Disruption of Intracellular Calcium Homeostasis as a Therapeutic Target Against Trypanosoma cruzi
Abstract
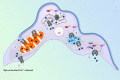
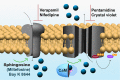
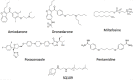
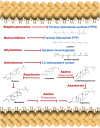
There is no effective cure for Chagas disease, which is caused by infection with the arthropod-borne parasite, Trypanosoma cruzi. In the search for new drugs to treat Chagas disease, potential therapeutic targets have been identified by exploiting the differences between the mechanisms involved in intracellular Ca2+ homeostasis, both in humans and in trypanosomatids. In the trypanosomatid, intracellular Ca2+ regulation requires the concerted action of three intracellular organelles, the endoplasmic reticulum, the single unique mitochondrion, and the acidocalcisomes. The single unique mitochondrion and the acidocalcisomes also play central roles in parasite bioenergetics. At the parasite plasma membrane, a Ca2+--ATPase (PMCA) with significant differences from its human counterpart is responsible for Ca2+ extrusion; a distinctive sphingosine-activated Ca2+ channel controls Ca2+ entrance to the parasite interior. Several potential anti-trypansosomatid drugs have been demonstrated to modulate one or more of these mechanisms for Ca2+ regulation. The antiarrhythmic agent amiodarone and its derivatives have been shown to exert trypanocidal effects through the disruption of parasite Ca2+ homeostasis. Similarly, the amiodarone-derivative dronedarone disrupts Ca2+ homeostasis in T. cruzi epimastigotes, collapsing the mitochondrial membrane potential (ΔΨm), and inducing a large increase in the intracellular Ca2+ concentration ([Ca2+]i) from this organelle and from the acidocalcisomes in the parasite cytoplasm. The same general mechanism has been demonstrated for SQ109, a new anti-tuberculosis drug with potent trypanocidal effect. Miltefosine similarly induces a large increase in the [Ca2+]i acting on the sphingosine-activated Ca2+ channel, the mitochondrion and acidocalcisomes. These examples, in conjunction with other evidence we review herein, strongly support targeting Ca2+ homeostasis as a strategy against Chagas disease.
Keywords: calcium; new drugs candidates; signaling; therapeutic target; trypanosomatids.
Copyright © 2020 Benaim, Paniz-Mondolfi, Sordillo and Martinez-Sotillo.
Figures
References
-
- Adesse D., Meirelles Azzam E., Meirelles M. N., Urbina J. A., Garzoni L. R. (2011). Amiodarone inhibits Trypanosoma cruzi infection and iromotes cardiac cell recovery with gap junction and cytoskeleton reassembly in vitro. Antimicrob. Agents Chemother. 55, 203–210. 10.1128/AAC.01129-10 - DOI - PMC - PubMed
-
- Benaim G., Cervino V., Hermoso T., Felibertt P., Laurentin A. (1993a). Intracellular calcium homeostasis in Leishmania mexicana. Identification and characterization of a plasma membrane calmodulin-dependent Ca2+-ATPase. Biol. Res. 26, 141–150. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

