Roles of pattern recognition receptors in diabetic nephropathy
- PMID: 32133797
- PMCID: PMC7086007
- DOI: 10.1631/jzus.B1900490
Roles of pattern recognition receptors in diabetic nephropathy
Abstract
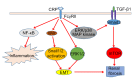
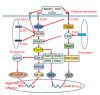
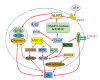
Diabetic nephropathy (DN) is currently the most common complication of diabetes. It is considered to be one of the leading causes of end-stage renal disease (ESRD) and affects many diabetic patients. The pathogenesis of DN is extremely complex and has not yet been clarified; however, in recent years, increasing evidence has shown the important role of innate immunity in DN pathogenesis. Pattern recognition receptors (PRRs) are important components of the innate immune system and have a significant impact on the occurrence and development of DN. In this review, we classify PRRs into secretory, endocytic, and signal transduction PRRs according to the relationship between the PRRs and subcellular compartments. PRRs can recognize related pathogen-associated molecular patterns (PAMPs) and danger-associated molecular patterns (DAMPs), thus triggering a series of inflammatory responses, promoting renal fibrosis, and finally causing renal impairment. In this review, we describe the proposed role of each type of PRRs in the development and progression of DN.
Keywords: Diabetic nephropathy; Innate immunity; Pattern recognition receptor; Pathogenesis.
Conflict of interest statement
This article does not contain any studies with human or animal subjects performed by any of the authors.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

