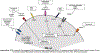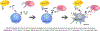Monoclonal antibodies in multiple myeloma: Current and emerging targets and mechanisms of action
- PMID: 32139009
- PMCID: PMC7060936
- DOI: 10.1016/j.beha.2020.101143
Monoclonal antibodies in multiple myeloma: Current and emerging targets and mechanisms of action
Abstract
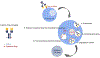
The recent development of monoclonal antibodies (mAbs) has revolutionized the treatment armamentarium for multiple myeloma. The success of daratumumab and elotuzumab in relapsed/refractory patients, has generated tremendous enthusiasm for mAbs in this disease. Combination treatment with other anti-myeloma treatment modalities and clinical evaluation in newly diagnosed patients are expected to fundamentally change the natural history of the disease. Advances in biopharmaceutical engineering together with a robust interest in novel mAb-derivatives, including antibody drug conjugates and poly-specific antibodies are the next rapidly approaching treatment frontier in multiple myeloma. In this review, we comprehensively outline the currently available evidence and the future landscape of mAbs and mAb-derivative therapies in multiple myeloma.
Keywords: Antibody drug conjugate; Bispecific antibody; Monoclonal antibody; Multiple myeloma.
Copyright © 2020 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of competing interest A.M.L. reports consultancy for Genmab, Bristol-Myers Squibb, and Takeda, honoraria from Genmab, Bristol-Myers Squibb, and Takeda, research grants from Genentech, Bristol-Myers Squibb, and Janssen, royalties from Serametrix. K.W. and B.W. declare no conflict of interests.
Figures
References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin 2019;69(1):7–34. - PubMed
-
- Lokhorst HM, Plesner T, Laubach JP, Nahi H, Gimsing P, Hansson M, et al. Targeting CD38 with Daratumumab Monotherapy in Multiple Myeloma. N Engl J Med 2015;373(13):1207–19. - PubMed
-
- Lonial S, Weiss BM, Usmani SZ, Singhal S, Chari A, Bahlis NJ, et al. Daratumumab monotherapy in patients with treatment-refractory multiple myeloma (SIRIUS): an open-label, randomised, phase 2 trial. Lancet 2016;387(10027):1551–60. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical