Omalizumab for Aspirin Hypersensitivity and Leukotriene Overproduction in Aspirin-exacerbated Respiratory Disease. A Randomized Controlled Trial
- PMID: 32142372
- PMCID: PMC7301746
- DOI: 10.1164/rccm.201906-1215OC
Omalizumab for Aspirin Hypersensitivity and Leukotriene Overproduction in Aspirin-exacerbated Respiratory Disease. A Randomized Controlled Trial
Abstract
Rationale: Aspirin-exacerbated respiratory disease is characterized by severe asthma, nonsteroidal antiinflammatory drug hypersensitivity, nasal polyposis, and leukotriene overproduction. Systemic corticosteroid therapy does not completely suppress lifelong aspirin hypersensitivity. Omalizumab efficacy against aspirin-exacerbated respiratory disease has not been investigated in a randomized manner.Objectives: To evaluate omalizumab efficacy against aspirin hypersensitivity, leukotriene E4 overproduction, and symptoms during an oral aspirin challenge in patients with aspirin-exacerbated respiratory disease using a randomized design.Methods: We performed a double-blind, randomized, crossover, placebo-controlled, single-center study at Sagamihara National Hospital between August 2015 and December 2016. Atopic patients (20-79 yr old) with aspirin-exacerbated respiratory disease diagnosed by systemic aspirin challenge were randomized (1:1) to a 3-month treatment with omalizumab or placebo, followed by a >18-week washout period (crossover design). The primary endpoint was the difference in area under logarithm level of urinary leukotriene E4 concentration versus time curve in the intent-to-treat population during an oral aspirin challenge.Measurements and Main Results: Sixteen patients completed the study and were included in the analysis. The area under the logarithm level of urinary leukotriene E4 concentration versus time curve during an oral aspirin challenge was significantly lower in the omalizumab phase (median [interquartile range], 51.1 [44.5-59.8]) than in the placebo phase (80.8 [interquartile range, 65.4-87.8]) (P < 0.001). Ten of 16 patients (62.5%) developed oral aspirin tolerance up to cumulative doses of 930 mg in the omalizumab phase (P < 0.001).Conclusions: Omalizumab treatment inhibited urinary leukotriene E4 overproduction and upper/lower respiratory tract symptoms during an oral aspirin challenge, resulting in aspirin tolerance in 62.5% of the patients with aspirin-exacerbated respiratory disease.
Keywords: aspirin hypersensitivity; aspirin tolerance; aspirin-exacerbated respiratory disease; leukotriene E4; omalizumab.
Figures
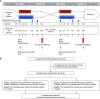
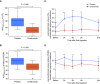


Comment in
-
Does IgE Have a Role in Aspirin-exacerbated Respiratory Disease?Am J Respir Crit Care Med. 2020 Jun 15;201(12):1459-1460. doi: 10.1164/rccm.202003-0783ED. Am J Respir Crit Care Med. 2020. PMID: 32286851 Free PMC article. No abstract available.
References
-
- Laidlaw TM, Boyce JA. Aspirin-exacerbated respiratory disease: new prime suspects. N Engl J Med. 2016;374:484–488. - PubMed
-
- White AA, Stevenson DD. Aspirin-exacerbated respiratory disease. N Engl J Med. 2018;379:1060–1070. - PubMed
-
- Kowalski ML, Agache I, Bavbek S, Bakirtas A, Blanca M, Bochenek G, et al. Diagnosis and management of NSAID-Exacerbated Respiratory Disease (N-ERD)-a EAACI position paper. Allergy. 2019;74:28–39. - PubMed
-
- Christie PE, Tagari P, Ford-Hutchinson AW, Charlesson S, Chee P, Arm JP, et al. Urinary leukotriene E4 concentrations increase after aspirin challenge in aspirin-sensitive asthmatic subjects. Am Rev Respir Dis. 1991;143:1025–1029. - PubMed
-
- Johns CB, Laidlaw TM. Elevated total serum IgE in nonatopic patients with aspirin-exacerbated respiratory disease. Am J Rhinol Allergy. 2014;28:287–289. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

