A highly responsive pyruvate sensor reveals pathway-regulatory role of the mitochondrial pyruvate carrier MPC
- PMID: 32142409
- PMCID: PMC7077990
- DOI: 10.7554/eLife.53917
A highly responsive pyruvate sensor reveals pathway-regulatory role of the mitochondrial pyruvate carrier MPC
Abstract
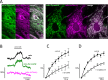
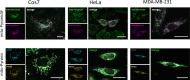
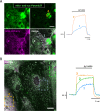
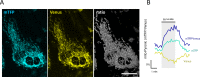
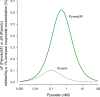
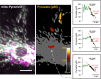
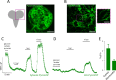
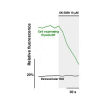
Mitochondria generate ATP and building blocks for cell growth and regeneration, using pyruvate as the main substrate. Here we introduce PyronicSF, a user-friendly GFP-based sensor of improved dynamic range that enables real-time subcellular quantitation of mitochondrial pyruvate transport, concentration and flux. We report that cultured mouse astrocytes maintain mitochondrial pyruvate in the low micromolar range, below cytosolic pyruvate, which means that the mitochondrial pyruvate carrier MPC is poised to exert ultrasensitive control on the balance between respiration and anaplerosis/gluconeogenesis. The functionality of the sensor in living tissue is demonstrated in the brain of Drosophila melanogaster larvae. Mitochondrial subpopulations are known to coexist within a given cell, which differ in their morphology, mobility, membrane potential, and vicinity to other organelles. The present tool can be used to investigate how mitochondrial diversity relates to metabolism, to study the role of MPC in disease, and to screen for small-molecule MPC modulators.
Keywords: D. melanogaster; cell biology; energy metabolism; genetically-encoded sensor; mitochondria; mouse; pyruvate; transport-stop protocol.
© 2020, Arce-Molina et al.
Conflict of interest statement
RA, FC, PS, AG, KA, SS, LB, AS No competing interests declared
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

