Two small, cysteine-rich and cationic antifungal proteins from Penicillium chrysogenum: A comparative study of PAF and PAFB
- PMID: 32142818
- PMCID: PMC7138148
- DOI: 10.1016/j.bbamem.2020.183246
Two small, cysteine-rich and cationic antifungal proteins from Penicillium chrysogenum: A comparative study of PAF and PAFB
Abstract
The filamentous fungus Penicillium chrysogenum Q176 secretes the antimicrobial proteins (AMPs) PAF and PAFB, which share a compact disulfide-bond mediated, β-fold structure rendering them highly stable. These two AMPs effectively inhibit the growth of human pathogenic fungi in micromolar concentrations and exhibit antiviral potential without causing cytotoxic effects on mammalian cells in vitro and in vivo. The antifungal mechanism of action of both AMPs is closely linked to - but not solely dependent on - the lipid composition of the fungal cell membrane and requires a strictly regulated protein uptake into the cell, indicating that PAF and PAFB are not canonical membrane active proteins. Variations in their antifungal spectrum and their killing dynamics point towards a divergent mode of action related to their physicochemical properties and surface charge distribution. In this review, we relate characteristic features of PAF and PAFB to the current knowledge about other AMPs of different sources. In addition, we present original data that have never been published before to substantiate our assumptions and provide evidences that help to explain and understand better the mechanistic function of PAF and PAFB. Finally, we underline the promising potential of PAF and PAFB as future antifungal therapeutics.
Keywords: Antimicrobial proteins and peptides; Apoptosis; Endocytosis; Fungal membrane lipids; Penicillium chrysogenum; β-Fold structure; γ-Core.
Copyright © 2020 The Author(s). Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





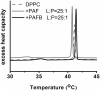

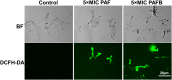
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

