Notch-mediated lateral induction is necessary to maintain vestibular prosensory identity during inner ear development
- PMID: 32147304
- PMCID: PMC7261482
- DOI: 10.1016/j.ydbio.2020.02.015
Notch-mediated lateral induction is necessary to maintain vestibular prosensory identity during inner ear development
Abstract
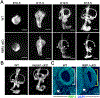
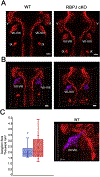
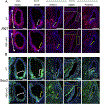
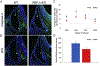
The five vestibular organs of the inner ear derive from patches of prosensory cells that express the transcription factor SOX2 and the Notch ligand JAG1. Previous work suggests that JAG1-mediated Notch signaling is both necessary and sufficient for prosensory formation and that the separation of developing prosensory patches is regulated by LMX1a, which antagonizes Notch signaling. We used an inner ear-specific deletion of the Rbpjκ gene in which Notch signaling is progressively lost from the inner ear to show that Notch signaling, is continuously required for the maintenance of prosensory fate. Loss of Notch signaling in prosensory patches causes them to shrink and ultimately disappear. We show this loss of prosensory fate is not due to cell death, but rather to the conversion of prosensory tissue into non-sensory tissue that expresses LMX1a. Notch signaling is therefore likely to stabilize, rather than induce prosensory fate.
Keywords: Development; Inner ear; Notch; Otic; Sensory; Vestibular.
Copyright © 2020 Elsevier Inc. All rights reserved.
Figures



Similar articles
-
Notch signalling is needed to maintain, but not to initiate, the formation of prosensory patches in the chick inner ear.Development. 2007 Jun;134(12):2369-78. doi: 10.1242/dev.001842. Development. 2007. PMID: 17537801
-
Jagged 1 regulates the restriction of Sox2 expression in the developing chicken inner ear: a mechanism for sensory organ specification.Development. 2011 Feb;138(4):735-44. doi: 10.1242/dev.060657. Development. 2011. PMID: 21266409
-
The Notch ligand JAG1 is required for sensory progenitor development in the mammalian inner ear.PLoS Genet. 2006 Jan;2(1):e4. doi: 10.1371/journal.pgen.0020004. Epub 2006 Jan 13. PLoS Genet. 2006. PMID: 16410827 Free PMC article.
-
Patterning and cell fate in the inner ear: a case for Notch in the chicken embryo.Dev Growth Differ. 2013 Jan;55(1):96-112. doi: 10.1111/dgd.12016. Epub 2012 Dec 17. Dev Growth Differ. 2013. PMID: 23252974 Review.
-
Notch Signalling: The Multitask Manager of Inner Ear Development and Regeneration.Adv Exp Med Biol. 2020;1218:129-157. doi: 10.1007/978-3-030-34436-8_8. Adv Exp Med Biol. 2020. PMID: 32060875 Review.
Cited by
-
A single-cell level comparison of human inner ear organoids with the human cochlea and vestibular organs.Cell Rep. 2023 Jun 27;42(6):112623. doi: 10.1016/j.celrep.2023.112623. Epub 2023 Jun 7. Cell Rep. 2023. PMID: 37289589 Free PMC article.
-
Notch1 is required to maintain supporting cell identity and vestibular function during maturation of the mammalian balance organs.bioRxiv [Preprint]. 2024 Jun 21:2024.06.21.600098. doi: 10.1101/2024.06.21.600098. bioRxiv. 2024. Update in: J Neurosci. 2025 Feb 26;45(9):e1365242024. doi: 10.1523/JNEUROSCI.1365-24.2024. PMID: 38948821 Free PMC article. Updated. Preprint.
-
Recent advances in in situ Notch signaling measurement.Front Cell Dev Biol. 2023 Jul 28;11:1244105. doi: 10.3389/fcell.2023.1244105. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37576594 Free PMC article. Review.
-
The Notch Ligand Jagged1 Is Required for the Formation, Maintenance, and Survival of Hensen's Cells in the Mouse Cochlea.J Neurosci. 2020 Dec 2;40(49):9401-9413. doi: 10.1523/JNEUROSCI.1192-20.2020. Epub 2020 Oct 30. J Neurosci. 2020. PMID: 33127852 Free PMC article.
-
Molecular Interplay in Cardiac Fibrosis: Exploring the Functions of RUNX2, BMP2, and Notch.Rev Cardiovasc Med. 2024 Oct 16;25(10):368. doi: 10.31083/j.rcm2510368. eCollection 2024 Oct. Rev Cardiovasc Med. 2024. PMID: 39484128 Free PMC article. Review.
References
-
- Abelló G, Khatri S, Gir aldez F, Alsina B, 2007. Early regionalization of the otic placode and its regulation by the Notch signaling pathway. Mech. Dev 124, 631–645. - PubMed
-
- Abelló G, Khatri S, Radosevic M, Scotting PJ, Gir aldez F, Alsina B, 2010. Independent regulation of Sox3 and Lmx1b by FGF and BMP signaling influences the neurogenic and non-neurogenic domains in the chick otic placode. Dev. Biol 339, 166–178. - PubMed
-
- Adam J, Myat A, Le Roux I, Eddison M, Henrique D, Ish-Horowicz D, Lewis J, 1998. Cell fate choices and the expression of Notch, Delta and Serrate homologues in the chick inner ear: parallels with Drosophila sense-organ development. Development 125, 4645–4654. - PubMed
-
- Alsina B, Giraldez F, Pujades C, 2009. Patterning and cell fate in ear development. Int. J. Dev. Biol 53, 1503–1513. - PubMed
-
- Ambler CA, Watt FM, 2007. Expression of Notch pathway genes in mammalian epidermis and modulation by beta-catenin. Dev. Dynam 236, 1595–1601. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

