Neonatal Hypocalcemic Seizures in Offspring of a Mother With Familial Hypocalciuric Hypercalcemia Type 1 (FHH1)
- PMID: 32150253
- PMCID: PMC7096312
- DOI: 10.1210/clinem/dgaa111
Neonatal Hypocalcemic Seizures in Offspring of a Mother With Familial Hypocalciuric Hypercalcemia Type 1 (FHH1)
Abstract
Context: Familial hypocalciuric hypercalcemia type 1 (FHH1) is caused by loss-of-function mutations of the calcium-sensing receptor (CaSR) and is considered a benign condition associated with mild-to-moderate hypercalcemia. However, the children of parents with FHH1 can develop a variety of disorders of calcium homeostasis in infancy.
Objective: The objective of this work is to characterize the range of calcitropic phenotypes in the children of a mother with FHH1.
Methods: A 3-generation FHH kindred was assessed by clinical, biochemical, and mutational analysis following informed consent.
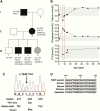
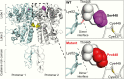
Results: The FHH kindred comprised a hypercalcemic man and his daughter who had hypercalcemia and hypocalciuria, and her 4 children, 2 of whom had asymptomatic hypercalcemia, 1 was normocalcemic, and 1 suffered from transient neonatal hypocalcemia and seizures. The hypocalcemic infant had a serum calcium of 1.57 mmol/L (6.28 mg/dL); normal, 2.0 to 2.8 mmol/L (8.0-11.2 mg/dL) and parathyroid hormone of 2.2 pmol/L; normal 1.0 to 9.3 pmol/L, and required treatment with intravenous calcium gluconate infusions. A novel heterozygous p.Ser448Pro CaSR variant was identified in the hypercalcemic individuals, but not the children with hypocalcemia or normocalcemia. Three-dimensional modeling predicted the p.Ser448Pro variant to disrupt a hydrogen bond interaction within the CaSR extracellular domain. The variant Pro448 CaSR, when expressed in HEK293 cells, significantly impaired CaSR-mediated intracellular calcium mobilization and mitogen-activated protein kinase responses following stimulation with extracellular calcium, thereby demonstrating it to represent a loss-of-function mutation.
Conclusions: Thus, children of a mother with FHH1 can develop hypercalcemia or transient neonatal hypocalcemia, depending on the underlying inherited CaSR mutation, and require investigations for serum calcium and CaSR mutations in early childhood.
Keywords: calcium-sensing receptor; hypercalcemia; hypocalcemia; hypoparathyroidism; loss-of-function; mutation.
© Endocrine Society 2020.
Figures


Similar articles
-
Heterozygous Mutation (Q459R) in the Calcium-Sensing Receptor Gene Causes Familial Hypocalciuric Hypercalcemia 1 (FHH1).J Clin Endocrinol Metab. 2020 Apr 1;105(4):dgz205. doi: 10.1210/clinem/dgz205. J Clin Endocrinol Metab. 2020. PMID: 32160303
-
Familial Hypocalciuric Hypercalcemia Type 1 and Autosomal-Dominant Hypocalcemia Type 1: Prevalence in a Large Healthcare Population.Am J Hum Genet. 2020 Jun 4;106(6):734-747. doi: 10.1016/j.ajhg.2020.04.006. Epub 2020 May 7. Am J Hum Genet. 2020. PMID: 32386559 Free PMC article.
-
A novel germline inactivating mutation in the CASR gene in an Italian kindred affected by familial hypocalciuric hypercalcemia.Eur J Endocrinol. 2012 May;166(5):933-40. doi: 10.1530/EJE-11-0953. Epub 2012 Feb 7. Eur J Endocrinol. 2012. PMID: 22315359
-
Mutations of the calcium-sensing receptor (CASR) in familial hypocalciuric hypercalcemia, neonatal severe hyperparathyroidism, and autosomal dominant hypocalcemia.Hum Mutat. 2000 Oct;16(4):281-96. doi: 10.1002/1098-1004(200010)16:4<281::AID-HUMU1>3.0.CO;2-A. Hum Mutat. 2000. PMID: 11013439 Review.
-
Rare diseases caused by abnormal calcium sensing and signalling.Endocrine. 2021 Mar;71(3):611-617. doi: 10.1007/s12020-021-02620-5. Epub 2021 Feb 2. Endocrine. 2021. PMID: 33528764 Free PMC article. Review.
Cited by
-
Evolutionary history of calcium-sensing receptors unveils hyper/hypocalcemia-causing mutations.PLoS Comput Biol. 2024 Nov 12;20(11):e1012591. doi: 10.1371/journal.pcbi.1012591. eCollection 2024 Nov. PLoS Comput Biol. 2024. PMID: 39531485 Free PMC article.
-
Neonatal hypocalcaemic seizures unmasking asymptomatic maternal primary hyperparathyroidism.BMJ Case Rep. 2024 Feb 15;17(2):e258134. doi: 10.1136/bcr-2023-258134. BMJ Case Rep. 2024. PMID: 38359958
-
Spectrum of germline AIRE mutations causing APS-1 and familial hypoparathyroidism.Eur J Endocrinol. 2022 May 24;187(1):111-122. doi: 10.1530/EJE-21-0730. Eur J Endocrinol. 2022. PMID: 35521792 Free PMC article.
-
Genetics of monogenic disorders of calcium and bone metabolism.Clin Endocrinol (Oxf). 2022 Oct;97(4):483-501. doi: 10.1111/cen.14644. Epub 2021 Dec 21. Clin Endocrinol (Oxf). 2022. PMID: 34935164 Free PMC article. Review.
-
Clinical, Biochemical, and Genetic Profile of an Indian Kindred with Type 1 Familial Hypocalciuric Hypercalcemia.Indian J Endocrinol Metab. 2021 Sep-Oct;25(5):462-465. doi: 10.4103/ijem.ijem_349_21. Epub 2022 Jan 12. Indian J Endocrinol Metab. 2021. PMID: 35300448 Free PMC article. No abstract available.
References
-
- Conigrave AD, Ward DT. Calcium-sensing receptor (CaSR): pharmacological properties and signaling pathways. Best Pract Res Clin Endocrinol Metab. 2013;27(3):315–331. - PubMed
-
- Murphy H, Patrick J, Báez-Irizarry E, et al. . Neonatal severe hyperparathyroidism caused by homozygous mutation in CASR: a rare cause of life-threatening hypercalcemia. Eur J Med Genet. 2016;59(4):227–231. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

