Conditional KCa3.1-transgene induction in murine skin produces pruritic eczematous dermatitis with severe epidermal hyperplasia and hyperkeratosis
- PMID: 32150577
- PMCID: PMC7062274
- DOI: 10.1371/journal.pone.0222619
Conditional KCa3.1-transgene induction in murine skin produces pruritic eczematous dermatitis with severe epidermal hyperplasia and hyperkeratosis
Abstract
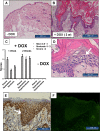
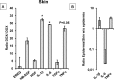
Ion channels have recently attracted attention as potential mediators of skin disease. Here, we explored the consequences of genetically encoded induction of the cell volume-regulating Ca2+-activated KCa3.1 channel (Kcnn4) for murine epidermal homeostasis. Doxycycline-treated mice harboring the KCa3.1+-transgene under the control of the reverse tetracycline-sensitive transactivator (rtTA) showed 800-fold channel overexpression above basal levels in the skin and solid KCa3.1-currents in keratinocytes. This overexpression resulted in epidermal spongiosis, progressive epidermal hyperplasia and hyperkeratosis, itch and ulcers. The condition was accompanied by production of the pro-proliferative and pro-inflammatory cytokines, IL-β1 (60-fold), IL-6 (33-fold), and TNFα (26-fold) in the skin. Treatment of mice with the KCa3.1-selective blocker, Senicapoc, significantly suppressed spongiosis and hyperplasia, as well as induction of IL-β1 (-88%) and IL-6 (-90%). In conclusion, KCa3.1-induction in the epidermis caused expression of pro-proliferative cytokines leading to spongiosis, hyperplasia and hyperkeratosis. This skin condition resembles pathological features of eczematous dermatitis and identifies KCa3.1 as a regulator of epidermal homeostasis and spongiosis, and as a potential therapeutic target.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Repurposing the KCa3.1 Blocker Senicapoc for Ischemic Stroke.Transl Stroke Res. 2024 Jun;15(3):518-532. doi: 10.1007/s12975-023-01152-6. Epub 2023 Apr 24. Transl Stroke Res. 2024. PMID: 37088858 Free PMC article.
-
KCa3.1 Transgene Induction in Murine Intestinal Epithelium Causes Duodenal Chyme Accumulation and Impairs Duodenal Contractility.Int J Mol Sci. 2019 Mar 8;20(5):1193. doi: 10.3390/ijms20051193. Int J Mol Sci. 2019. PMID: 30857243 Free PMC article.
-
Expression of T-cell KV1.3 potassium channel correlates with pro-inflammatory cytokines and disease activity in ulcerative colitis.J Crohns Colitis. 2014 Nov;8(11):1378-91. doi: 10.1016/j.crohns.2014.04.003. Epub 2014 May 3. J Crohns Colitis. 2014. PMID: 24793818 Free PMC article.
-
Intermediate conductance calcium-activated potassium channel (KCa3.1) in cancer: Emerging roles and therapeutic potentials.Biochem Pharmacol. 2024 Dec;230(Pt 1):116573. doi: 10.1016/j.bcp.2024.116573. Epub 2024 Oct 11. Biochem Pharmacol. 2024. PMID: 39396649 Review.
-
Senicapoc: Repurposing a Drug to Target Microglia KCa3.1 in Stroke.Neurochem Res. 2017 Sep;42(9):2639-2645. doi: 10.1007/s11064-017-2223-y. Epub 2017 Mar 31. Neurochem Res. 2017. PMID: 28364331 Free PMC article. Review.
Cited by
-
Barium and psoriasis: a mini-review and hypothesis linking environmental exposures to ion channel modulation.Front Med (Lausanne). 2025 Jun 18;12:1585525. doi: 10.3389/fmed.2025.1585525. eCollection 2025. Front Med (Lausanne). 2025. PMID: 40606452 Free PMC article. Review.
-
Pathophysiological Roles of Ion Channels in Epidermal Cells, Immune Cells, and Sensory Neurons in Psoriasis.Int J Mol Sci. 2024 Feb 27;25(5):2756. doi: 10.3390/ijms25052756. Int J Mol Sci. 2024. PMID: 38474002 Free PMC article. Review.
References
-
- Oliván-Viguera A, Garcia-Otin AL, Lozano-Gerona J, Abarca-Lachen E, Garcia-Malinis AJ, Hamilton KL, et al. Pharmacological activation of TRPV4 produces immediate cell damage and induction of apoptosis in human melanoma cells and HaCaT keratinocytes. PLoS One. 2018;13(1): e0190307 10.1371/journal.pone.0190307 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

