mTOR and STAT3 Pathway Hyper-Activation is Associated with Elevated Interleukin-6 Levels in Patients with Shwachman-Diamond Syndrome: Further Evidence of Lymphoid Lineage Impairment
- PMID: 32150944
- PMCID: PMC7139896
- DOI: 10.3390/cancers12030597
mTOR and STAT3 Pathway Hyper-Activation is Associated with Elevated Interleukin-6 Levels in Patients with Shwachman-Diamond Syndrome: Further Evidence of Lymphoid Lineage Impairment
Abstract
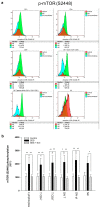
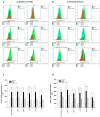
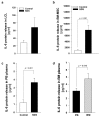
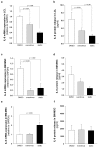
Shwachman-Diamond syndrome (SDS) is a rare inherited bone marrow failure syndrome, resulting in neutropenia and a risk of myeloid neoplasia. A mutation in a ribosome maturation factor accounts for almost all of the cases. Lymphoid involvement in SDS has not been well characterized. We recently reported that lymphocyte subpopulations are reduced in SDS patients. We have also shown that the mTOR-STAT3 pathway is hyper-activated in SDS myeloid cell populations. Here we show that mTOR-STAT3 signaling is markedly upregulated in the lymphoid compartment of SDS patients. Furthermore, our data reveal elevated IL-6 levels in cellular supernatants obtained from lymphoblasts, bone marrow mononuclear and mesenchymal stromal cells, and plasma samples obtained from a cohort of 10 patients. Of note, everolimus-mediated inhibition of mTOR signaling is associated with basal state of phosphorylated STAT3. Finally, inhibition of mTOR-STAT3 pathway activation leads to normalization of IL-6 expression in SDS cells. Altogether, our data strengthen the hypothesis that SDS affects both lymphoid and myeloid blood compartment and suggest everolimus as a potential therapeutic agent to reduce excessive mTOR-STAT3 activation in SDS.
Keywords: Bone Marrow Failure Syndromes; STAT3; lymphocytes; mTOR.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Constitutive systemic inflammation in Shwachman-Diamond Syndrome.Mol Med. 2025 Feb 28;31(1):81. doi: 10.1186/s10020-025-01133-5. Mol Med. 2025. PMID: 40021961 Free PMC article.
-
New insights into the Shwachman-Diamond Syndrome-related haematological disorder: hyper-activation of mTOR and STAT3 in leukocytes.Sci Rep. 2016 Sep 23;6:33165. doi: 10.1038/srep33165. Sci Rep. 2016. PMID: 27658964 Free PMC article.
-
Ataluren-driven restoration of Shwachman-Bodian-Diamond syndrome protein function in Shwachman-Diamond syndrome bone marrow cells.Am J Hematol. 2018 Aug;93(4):527-536. doi: 10.1002/ajh.25025. Epub 2018 Feb 9. Am J Hematol. 2018. PMID: 29285795
-
Shwachman-Diamond syndrome.Semin Hematol. 2002 Apr;39(2):95-102. doi: 10.1053/shem.2002.31915. Semin Hematol. 2002. PMID: 11957191 Review.
-
Mechanisms of leukemic transformation in congenital neutropenia.Curr Opin Hematol. 2019 Jan;26(1):34-40. doi: 10.1097/MOH.0000000000000479. Curr Opin Hematol. 2019. PMID: 30431463 Free PMC article. Review.
Cited by
-
Expression of JAK/STAT Signaling Proteins at Diagnosis and Remission in Patients with Acute Myeloid Leukemia.Oman Med J. 2024 May 30;39(3):e633. doi: 10.5001/omj.2024.80. eCollection 2024 May. Oman Med J. 2024. PMID: 39497941 Free PMC article.
-
mTORC1 and STAT3 signaling are indispensable for in vitro TGFβ1-dependent three-dimensional (3D) tendon constructs.BMB Rep. 2025 Jun;58(6):257-263. doi: 10.5483/BMBRep.2024-0198. BMB Rep. 2025. PMID: 40495482 Free PMC article.
-
Distinct peripheral pro-inflammatory profile associated with tuberous sclerosis complex and epilepsy.Epilepsia. 2025 Apr;66(4):1288-1303. doi: 10.1111/epi.18261. Epub 2025 Jan 16. Epilepsia. 2025. PMID: 39817839 Free PMC article.
-
Inflammatory pathways and the bone marrow microenvironment in inherited bone marrow failure syndromes.Stem Cells. 2025 May 27;43(6):sxaf021. doi: 10.1093/stmcls/sxaf021. Stem Cells. 2025. PMID: 40296192 Review.
-
Constitutive systemic inflammation in Shwachman-Diamond Syndrome.Mol Med. 2025 Feb 28;31(1):81. doi: 10.1186/s10020-025-01133-5. Mol Med. 2025. PMID: 40021961 Free PMC article.
References
-
- Dror Y., Donadieu J., Koglmeier J., Dodge J., Toiviainen-Salo S., Makitie O., Kerr E., Zeidler C., Shimamura A., Shah N., et al. Draft consensus guidelines for diagnosis and treatment of shwachman-diamond syndrome. Ann. N Y Acad. Sci. 2011;1242:40–55. doi: 10.1111/j.1749-6632.2011.06349.x. - DOI - PubMed
-
- Kargas V., Castro-Hartmann P., Escudero-Urquijo N., Dent K., Hilcenko C., Sailer C., Zisser G., Marques-Carvalho M.J., Pellegrino S., Wawiórka L., et al. Mechanism of completion of peptidyltransferase centre assembly in eukaryotes. Elife. 2019;8:e44904. doi: 10.7554/eLife.44904. - DOI - PMC - PubMed
-
- Finch A.J., Hilcenko C., Basse N., Drynan L.F., Goyenechea B., Menne T.F., González Fernández A., Simpson P., D’Santos C.S., Arends M.J., et al. Uncoupling of gtp hydrolysis from eif6 release on the ribosome causes shwachman-diamond syndrome. Genes Dev. 2011;25:917–929. doi: 10.1101/gad.623011. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

