Structural basis of Fusarium myosin I inhibition by phenamacril
- PMID: 32163521
- PMCID: PMC7100991
- DOI: 10.1371/journal.ppat.1008323
Structural basis of Fusarium myosin I inhibition by phenamacril
Abstract
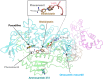
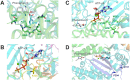
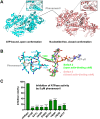
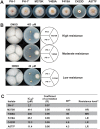
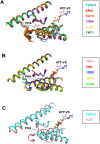
Fusarium is a genus of filamentous fungi that includes species that cause devastating diseases in major staple crops, such as wheat, maize, rice, and barley, resulting in severe yield losses and mycotoxin contamination of infected grains. Phenamacril is a novel fungicide that is considered environmentally benign due to its exceptional specificity; it inhibits the ATPase activity of the sole class I myosin of only a subset of Fusarium species including the major plant pathogens F. graminearum, F. asiaticum and F. fujikuroi. To understand the underlying mechanisms of inhibition, species specificity, and resistance mutations, we have determined the crystal structure of phenamacril-bound F. graminearum myosin I. Phenamacril binds in the actin-binding cleft in a new allosteric pocket that contains the central residue of the regulatory Switch 2 loop and that is collapsed in the structure of a myosin with closed actin-binding cleft, suggesting that pocket occupancy blocks cleft closure. We have further identified a single, transferable phenamacril-binding residue found exclusively in phenamacril-sensitive myosins to confer phenamacril selectivity.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Zheng Z, Hou Y, Cai Y, Zhang Y, Li Y, Zhou M. Whole-genome sequencing reveals that mutations in myosin-5 confer resistance to the fungicide phenamacril in Fusarium graminearum. Sci Rep. 2015;5:8248 10.1038/srep08248 https://www.nature.com/articles/srep08248#supplementary-information. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

