HSP Transcript and Protein Accumulation in Brassinosteroid Barley Mutants Acclimated to Low and High Temperatures
- PMID: 32164259
- PMCID: PMC7084868
- DOI: 10.3390/ijms21051889
HSP Transcript and Protein Accumulation in Brassinosteroid Barley Mutants Acclimated to Low and High Temperatures
Abstract
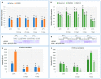
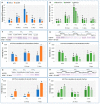
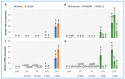
In temperature stress, the main role of heat-shock proteins (HSP) is to act as molecular chaperones for other cellular proteins. However, knowledge about the hormonal regulation of the production of the HSP is quite limited. Specifically, little is known about the role of the plant steroid hormones-brassinosteroids (BR)-in regulating the HSP expression. The aim of our study was to answer the question of how a BR deficit or disturbances in its signaling affect the accumulation of the HSP90, HSP70, HSP18, and HSP17 transcripts and protein in barley growing at 20 °C (control) and during the acclimation of plants at 5 °C and 27 °C. In barley, the temperature of plant growth modified the expression of HSPs. Furthermore, the BR-deficient mutants (mutations in the HvDWARF or HvCPD genes) and BR-signaling mutants (mutation in the HvBRI1 gene) were characterized by altered levels of the transcripts and proteins of the HSP group compared to the wild type. The BR-signaling mutant was characterized by a decreased level of the HSP transcripts and heat-shock proteins. In the BR-deficient mutants, there were temperature-dependent cases when the decreased accumulation of the HSP70 and HSP90 transcripts was connected to an increased accumulation of these HSP. The significance of changes in the accumulation of HSPs during acclimation at 27 °C and 5 °C is discussed in the context of the altered tolerance to more extreme temperatures of the studied mutants (i.e., heat stress and frost, respectively).
Keywords: HSP70; HSP90; acclimation process; brassinosteroids; small HSPs (sHSPs); temperature stress.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
-
- Awasthi R., Bhandari K., Nayyar H. Temperature stress and redox homeostasis in agricultural crops. Front. Environ. Sci. 2015;3:11. doi: 10.3389/fenvs.2015.00011. - DOI
-
- Hasanuzzaman M., Nahar K., Fujita M. Extreme temperature responses, oxidative stress and antioxidant defense in plants. In: Vahdati K., Leslie C., editors. Abiotic Stress Plant Responses Appllications in Agriculture. IntechOpen; London, UK: 2013. pp. 169–205.
-
- Howarth C.J. Molecular responses of plants to an increased incidence of heat shock. Plant Cell Environ. 1991;14:831–841. doi: 10.1111/j.1365-3040.1991.tb01446.x. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

