A half century of γ-aminobutyric acid
- PMID: 32166183
- PMCID: PMC7058221
- DOI: 10.1177/2398212819858249
A half century of γ-aminobutyric acid
Abstract
γ-aminobutyric acid has become one of the most widely known neurotransmitter molecules in the brain over the last 50 years, recognised for its pivotal role in inhibiting neural excitability. It emerged from studies of crustacean muscle and neurons before its significance to the mammalian nervous system was appreciated. Now, after five decades of investigation, we know that most neurons are γ-aminobutyric-acid-sensitive, it is a cornerstone of neural physiology and dysfunction to γ-aminobutyric acid signalling is increasingly documented in a range of neurological diseases. In this review, we briefly chart the neurodevelopment of γ-aminobutyric acid and its two major receptor subtypes: the γ-aminobutyric acidA and γ-aminobutyric acidB receptors, starting from the humble invertebrate origins of being an 'interesting molecule' acting at a single γ-aminobutyric acid receptor type, to one of the brain's most important neurochemical components and vital drug targets for major therapeutic classes of drugs. We document the period of molecular cloning and the explosive influence this had on the field of neuroscience and pharmacology up to the present day and the production of atomic γ-aminobutyric acidA and γ-aminobutyric acidB receptor structures. γ-Aminobutyric acid is no longer a humble molecule but the instigator of rich and powerful signalling processes that are absolutely vital for healthy brain function.
Keywords: GABA; GABAA receptor; GABAB receptor; inhibition.
© The Author(s) 2019.
Conflict of interest statement
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
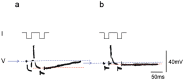
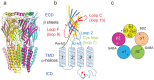


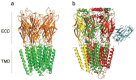
References
-
- Barber RP, Vaughn JE, Saito K, et al. (1978) GABAergic terminals are presynaptic to primary afferent terminals in the substantia gelatinosa of the rat spinal cord. Brain Research 141(1): 35–55. - PubMed
-
- Barnard EA, Darlison MG, Seeburg P. (1987) Molecular biology of the GABAA receptor: The receptor/channel super-family. Trends in Neurosciences 10(12): 502–509.
-
- Beaumont K, Chilton WS, Yamamura HI, et al. (1978) Muscimol binding in rat brain: Association with synaptic GABA receptors. Brain Research 148(1): 153–162. - PubMed
-
- Ben Ari Y. (2002) Excitatory actions of GABA during development: The nature of the nurture. Nature Reviews: Neuroscience 3(9): 728–739. - PubMed
-
- Ben Ari Y, Gaiarsa JL, Tyzio R, et al. (2007) GABA: A pioneer transmitter that excites immature neurons and generates primitive oscillations. Physiological Reviews 87(4): 1215–1284. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

