Streptococcus pneumoniae PepO promotes host anti-infection defense via autophagy in a Toll-like receptor 2/4 dependent manner
- PMID: 32172666
- PMCID: PMC7161686
- DOI: 10.1080/21505594.2020.1739411
Streptococcus pneumoniae PepO promotes host anti-infection defense via autophagy in a Toll-like receptor 2/4 dependent manner
Abstract
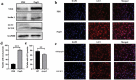
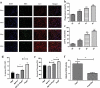
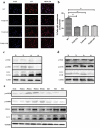
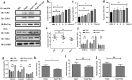
Macrophage is essential for host anti-bacterial defense by directly eliminating invading microbes and inducing a series of immune reactions. Here we identified a Streptococcus pneumoniae protein, PepO, as a TLR2/TLR4 bi-ligand. We found that PepO enhances macrophage unspecific phagocytosis and bactericidal activity, which is related to the induction of autophagy in macrophage, for the inhibition of autophagy significantly decreased the phagocytosis and bactericidal activity of PepO-treated macrophage. We confirmed that these effects of PepO are dependent on interacting with both TLR2 and TLR4. The tlr2 or tlr4 deficiency partially abolished the effect of PepO while tlr2/tlr4 deficiency abolished it completely. In vivo study demonstrated that PepO reduced the bacteria load in WT mice significantly, while the depletion of macrophage or tlr2/tlr4 deficiency abrogated the effect of PepO. Our findings suggested the therapeutic potential of PepO and provided experimental evidence for immunotherapy against infectious disease.
Keywords: Bacterial infection; autophagy; immunotherapy; macrophage; toll-like receptor.
Figures


Similar articles
-
Streptococcus pneumoniae Endopeptidase O (PepO) Elicits a Strong Innate Immune Response in Mice via TLR2 and TLR4 Signaling Pathways.Front Cell Infect Microbiol. 2016 Feb 29;6:23. doi: 10.3389/fcimb.2016.00023. eCollection 2016. Front Cell Infect Microbiol. 2016. PMID: 26973817 Free PMC article.
-
Purified Streptococcus pneumoniae Endopeptidase O (PepO) Enhances Particle Uptake by Macrophages in a Toll-Like Receptor 2- and miR-155-Dependent Manner.Infect Immun. 2017 Mar 23;85(4):e01012-16. doi: 10.1128/IAI.01012-16. Print 2017 Apr. Infect Immun. 2017. PMID: 28193634 Free PMC article.
-
Toll-Like receptor 2 (TLR2) and TLR9 play opposing roles in host innate immunity against Salmonella enterica serovar Typhimurium infection.Infect Immun. 2015 Apr;83(4):1641-9. doi: 10.1128/IAI.02870-14. Epub 2015 Feb 9. Infect Immun. 2015. PMID: 25667264 Free PMC article.
-
TLR2 and TLR4 mediated host immune responses in major infectious diseases: a review.Braz J Infect Dis. 2016 Mar-Apr;20(2):193-204. doi: 10.1016/j.bjid.2015.10.011. Epub 2016 Jan 14. Braz J Infect Dis. 2016. PMID: 26775799 Free PMC article. Review.
-
Mechanisms of TLR4-Mediated Autophagy and Nitroxidative Stress.Front Cell Infect Microbiol. 2021 Oct 22;11:766590. doi: 10.3389/fcimb.2021.766590. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 34746034 Free PMC article. Review.
Cited by
-
Nucleic acid sensing Toll-like receptors 3 and 9 play complementary roles in the development of bacteremia after nasal colonization associated with influenza co-infection.Exp Anim. 2024 Feb 14;73(1):50-60. doi: 10.1538/expanim.23-0001. Epub 2023 Aug 3. Exp Anim. 2024. PMID: 37532523 Free PMC article.
-
Intranasal trivalent candidate vaccine elicits broad humoral and cellular immunity against pneumococcal pneumonia.Front Cell Infect Microbiol. 2025 Jun 27;15:1563661. doi: 10.3389/fcimb.2025.1563661. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40654579 Free PMC article.
-
The Streptococcus virulence protein PepO triggers anti-tumor immune responses by reprograming tumor-associated macrophages in a mouse triple negative breast cancer model.Cell Biosci. 2023 Nov 4;13(1):198. doi: 10.1186/s13578-023-01153-w. Cell Biosci. 2023. PMID: 37925462 Free PMC article.
-
Streptococcus pneumoniae extracellular vesicles aggravate alveolar epithelial barrier disruption via autophagic degradation of OCLN (occludin).Autophagy. 2024 Jul;20(7):1577-1596. doi: 10.1080/15548627.2024.2330043. Epub 2024 Apr 21. Autophagy. 2024. PMID: 38497494 Free PMC article.
-
Molecular Mimicry Mapping in Streptococcus pneumoniae: Cues for Autoimmune Disorders and Implications for Immune Defense Activation.Pathogens. 2023 Jun 21;12(7):857. doi: 10.3390/pathogens12070857. Pathogens. 2023. PMID: 37513704 Free PMC article.
References
-
- Schaad UB. OM-85 BV, an immunostimulant in pediatric recurrent respiratory tract infections: a systematic review. World J Pediatr. 2010;6(1):5–12. - PubMed
-
- Steurer-Stey C, Bachmann LM, Steurer J, et al. Oral purified bacterial extracts in chronic bronchitis and COPD: systematic review. Chest. 2004;126(5):1645–1655. - PubMed
-
- Moresco EM, LaVine D, Beutler B. Toll-like receptors. Curr Biol. 2011;21(13):R488–93. - PubMed
-
- Hofmann U, Ertl G, Frantz S. Toll-like receptors as potential therapeutic targets in cardiac dysfunction. Expert Opin Ther Targets. 2011;15(6):753–765. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
