A Comprehensive Review of Alzheimer's Association with Related Proteins: Pathological Role and Therapeutic Significance
- PMID: 32172687
- PMCID: PMC7536827
- DOI: 10.2174/1570159X18666200203101828
A Comprehensive Review of Alzheimer's Association with Related Proteins: Pathological Role and Therapeutic Significance
Abstract
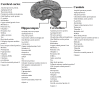
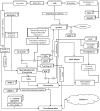
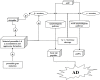
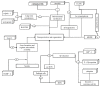
Alzheimer's is an insidious, progressive, chronic neurodegenerative disease which causes the devastation of neurons. Alzheimer's possesses complex pathologies of heterogeneous nature counting proteins as one major factor along with enzymes and mutated genes. Proteins such as amyloid precursor protein (APP), apolipoprotein E (ApoE), presenilin, mortalin, calbindin-D28K, creactive protein, heat shock proteins (HSPs), and prion protein are some of the chief elements in the foremost hypotheses of AD like amyloid-beta (Aβ) cascade hypothesis, tau hypothesis, cholinergic neuron damage, etc. Disturbed expression of these proteins results in synaptic dysfunction, cognitive impairment, memory loss, and neuronal degradation. On the therapeutic ground, attempts of developing anti-amyloid, anti-inflammatory, anti-tau therapies are on peak, having APP and tau as putative targets. Some proteins, e.g., HSPs, which ameliorate oxidative stress, calpains, which help in regulating synaptic plasticity, and calmodulin-like skin protein (CLSP) with its neuroprotective role are few promising future targets for developing anti-AD therapies. On diagnostic grounds of AD C-reactive protein, pentraxins, collapsin response mediator protein-2, and growth-associated protein-43 represent the future of new possible biomarkers for diagnosing AD. The last few decades were concentrated over identifying and studying protein targets of AD. Here, we reviewed the physiological/pathological roles and therapeutic significance of nearly all the proteins associated with AD that addresses putative as well as probable targets for developing effective anti-AD therapies.
Keywords: Alzheimer`s; neurodegeneration; pathological role; proteins; therapeutics.
Copyright© Bentham Science Publishers; For any queries, please email at epub@benthamscience.net.
Figures
Similar articles
-
Promising protein biomarkers in the early diagnosis of Alzheimer's disease.Metab Brain Dis. 2022 Aug;37(6):1727-1744. doi: 10.1007/s11011-021-00847-9. Epub 2022 Jan 11. Metab Brain Dis. 2022. PMID: 35015199 Review.
-
Key Peptides and Proteins in Alzheimer's Disease.Curr Protein Pept Sci. 2019;20(6):577-599. doi: 10.2174/1389203720666190103123434. Curr Protein Pept Sci. 2019. PMID: 30605056 Review.
-
Alzheimer's disease and amyloid: culprit or coincidence?Int Rev Neurobiol. 2012;102:277-316. doi: 10.1016/B978-0-12-386986-9.00011-9. Int Rev Neurobiol. 2012. PMID: 22748834 Review.
-
Amyloid cascade hypothesis: Pathogenesis and therapeutic strategies in Alzheimer's disease.Neuropeptides. 2015 Aug;52:1-18. doi: 10.1016/j.npep.2015.06.008. Epub 2015 Jul 2. Neuropeptides. 2015. PMID: 26149638 Review.
-
Alzheimer's disease.Subcell Biochem. 2012;65:329-52. doi: 10.1007/978-94-007-5416-4_14. Subcell Biochem. 2012. PMID: 23225010 Review.
Cited by
-
Protein Kinase C (PKC) in Neurological Health: Implications for Alzheimer's Disease and Chronic Alcohol Consumption.Brain Sci. 2024 May 29;14(6):554. doi: 10.3390/brainsci14060554. Brain Sci. 2024. PMID: 38928554 Free PMC article. Review.
-
The Proof-of-Concept of MBA121, a Tacrine-Ferulic Acid Hybrid, for Alzheimer's Disease Therapy.Int J Mol Sci. 2023 Jul 31;24(15):12254. doi: 10.3390/ijms241512254. Int J Mol Sci. 2023. PMID: 37569630 Free PMC article.
-
Symptomatic, Genetic, and Mechanistic Overlaps between Autism and Alzheimer's Disease.Biomolecules. 2021 Nov 4;11(11):1635. doi: 10.3390/biom11111635. Biomolecules. 2021. PMID: 34827633 Free PMC article. Review.
-
Role and application of CRISPR-Cas9 in the management of Alzheimer's disease.Ann Med Surg (Lond). 2024 Jan 5;86(3):1517-1521. doi: 10.1097/MS9.0000000000001692. eCollection 2024 Mar. Ann Med Surg (Lond). 2024. PMID: 38463115 Free PMC article. Review.
-
Unveiling the Molecular Footprint: Proteome-Based Biomarkers for Alzheimer's Disease.Proteomes. 2023 Oct 16;11(4):33. doi: 10.3390/proteomes11040033. Proteomes. 2023. PMID: 37873875 Free PMC article. Review.
References
-
- World Health Organization and Alzheimer’s Disease International Dementia: a public health priority. 2012.
-
- WHO Dementia Fact sheet. 2017 https://www.who.int/en/news-room/fact-sheets/detail/dementia
-
- Alzheimer’s & Related Disorders Society of India . The Dementia India Report: prevalence, impact, costs and services for Dementia: Executive Summary. New Delhi: ARDSI; 2010.
-
- Alzheimer’s Association 2018 Alzheimer’s disease Facts and Figures. Alzheimers Dement. 2018;14:367–429. doi: 10.1016/j.jalz.2018.02.001. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

