Kinetics of DNA Adducts and Abasic Site Formation in Tissues of Mice Treated with a Nitrogen Mustard
- PMID: 32174110
- PMCID: PMC7266082
- DOI: 10.1021/acs.chemrestox.0c00012
Kinetics of DNA Adducts and Abasic Site Formation in Tissues of Mice Treated with a Nitrogen Mustard
Abstract
Nitrogen mustards (NM) are an important class of chemotherapeutic drugs used in the treatment of malignant tumors. The accepted mechanism of action of NM is through the alkylation of DNA bases. NM-adducts block DNA replication in cancer cells by forming cytotoxic DNA interstrand cross-links. We previously characterized several adducts formed by reaction of bis(2-chloroethyl)ethylamine (NM) with calf thymus (CT) DNA and the MDA-MB-231 mammary tumor cell line. The monoalkylated N7-guanine (NM-G) adduct and its cross-link (G-NM-G) were major lesions. The cationic NM-G undergoes a secondary reaction through depurination to form an apurinic (AP) site or reacts with hydroxide to yield the stable ring-opened N5-substituted formamidopyrimidine (NM-Fapy-G) adduct. Both of these lesions are mutagenic and may contribute to secondary tumor development, a major clinical limitation of NM chemotherapy. We established a kinetic model with NM-treated female mice and measured the rates of formation and removal of NM-DNA adducts and AP sites. We employed liquid chromatography-mass spectrometry (LC-MS) to measure NM-G, G-NM-G, and NM-Fapy-G adducts in liver, lung, and spleen over 168 h. NM-G reached a maximum level within 6 h in all organs and then rapidly declined. The G-NM-G cross-link and NM-FapyG were more persistent with half-lives over three-times longer than NM-G. We quantified AP site lesions in the liver and showed that NM treatment increased AP site levels by 3.7-fold over the basal levels at 6 h. The kinetics of AP site repair closely followed the rate of removal of NM-G; however, AP sites remained 1.3-fold above basal levels 168 h post-treatment with NM. Our data provide new insights into NM-induced DNA damage and biological processing in vivo. The quantitative measurement of the spectrum of NM adducts and AP sites can serve as biomarkers in the design and assessment of the efficacy of novel chemotherapeutic regimens.
Figures

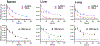


References
-
- Diethelm-Varela B, Ai Y, Liang DD, and Xue FT (2019) Nitrogen mustards as anticancer chemotherapies: Historic perspective, current developments and future trends. Curr. Top. Med. Chem 19, 691–712. - PubMed
-
- Einhorn J (1985) Nitrogen mustard: The origin of chemotherapy for cancer. Int. J. Radiat. Oncol. Biol. Phys 11, 1375–1378. - PubMed
-
- Emadi A, Jones RJ, and Brodsky RA (2009) Cyclophosphamide and cancer: golden anniversary. Nat. Rev. Clin. Oncol 6, 638–647. - PubMed
-
- Rajski SR, and Williams RM (1998) DNA cross-linking agents as antitumor drugs. Chem. Rev 98, 2723–2796. - PubMed
-
- McHugh PJ, Spanswick VJ, and Hartley JA (2001) Repair of DNA interstrand crosslinks: molecular mechanisms and clinical relevance. Lancet Oncol. 2, 483–490. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

