Disruption of Exocytosis in Sympathoadrenal Chromaffin Cells from Mouse Models of Neurodegenerative Diseases
- PMID: 32178443
- PMCID: PMC7139653
- DOI: 10.3390/ijms21061946
Disruption of Exocytosis in Sympathoadrenal Chromaffin Cells from Mouse Models of Neurodegenerative Diseases
Abstract
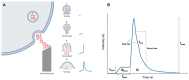
Synaptic disruption and altered neurotransmitter release occurs in the brains of patients and in murine models of neurodegenerative diseases (NDDs). During the last few years, evidence has accumulated suggesting that the sympathoadrenal axis is also affected as disease progresses. Here, we review a few studies done in adrenal medullary chromaffin cells (CCs), that are considered as the amplifying arm of the sympathetic nervous system; the sudden fast exocytotic release of their catecholamines-stored in noradrenergic and adrenergic cells-plays a fundamental role in the stress fight-or-flight response. Bulk exocytosis and the fine kinetics of single-vesicle exocytotic events have been studied in mouse models carrying a mutation linked to NDDs. For instance, in R6/1 mouse models of Huntington's disease (HD), mutated huntingtin is overexpressed in CCs; this causes decreased quantal secretion, smaller quantal size and faster kinetics of the exocytotic fusion pore, pore expansion, and closure. This was accompanied by decreased sodium current, decreased acetylcholine-evoked action potentials, and attenuated [Ca2+]c transients with faster Ca2+ clearance. In the SOD1G93A mouse model of amyotrophic lateral sclerosis (ALS), CCs exhibited secretory single-vesicle spikes with a slower release rate but higher exocytosis. Finally, in the APP/PS1 mouse model of Alzheimer's disease (AD), the stabilization, expansion, and closure of the fusion pore was faster, but the secretion was attenuated. Additionally, α-synuclein that is associated with Parkinson's disease (PD) decreases exocytosis and promotes fusion pore dilation in adrenal CCs. Furthermore, Huntington-associated protein 1 (HAP1) interacts with the huntingtin that, when mutated, causes Huntington's disease (HD); HAP1 reduces full fusion exocytosis by affecting vesicle docking and controlling fusion pore stabilization. The alterations described here are consistent with the hypothesis that central alterations undergone in various NDDs are also manifested at the peripheral sympathoadrenal axis to impair the stress fight-or-flight response in patients suffering from those diseases. Such alterations may occur: (i) primarily by the expression of mutated disease proteins in CCs; (ii) secondarily to stress adaptation imposed by disease progression and the limitations of patient autonomy.
Keywords: chromaffin cell; exocytosis; neurodegenerative diseases; sympathoadrenal axis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

