Niche Cells and Signals that Regulate Lung Alveolar Stem Cells In Vivo
- PMID: 32179507
- PMCID: PMC7706567
- DOI: 10.1101/cshperspect.a035717
Niche Cells and Signals that Regulate Lung Alveolar Stem Cells In Vivo
Erratum in
-
Corrigendum: Niche Cells and Signals that Regulate Lung Alveolar Stem Cells In Vivo.Cold Spring Harb Perspect Biol. 2020 May 1;12(5):a040303. doi: 10.1101/cshperspect.a040303. Cold Spring Harb Perspect Biol. 2020. PMID: 32358163 Free PMC article. No abstract available.
Abstract
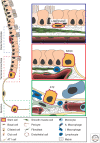
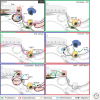
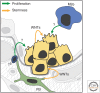
The distal lung is a honeycomb-like collection of delicate gas exchange sacs called alveoli lined by two interspersed epithelial cell types: the cuboidal, surfactant-producing alveolar type II (AT2) and the flat, gas-exchanging alveolar type I (AT1) cell. During aging, a subset of AT2 cells expressing the canonical Wnt target gene, Axin2, function as stem cells, renewing themselves while generating new AT1 and AT2 cells. Wnt activity endows AT2 cells with proliferative competency, enabling them to respond to activating cues, and simultaneously blocks AT2 to AT1 cell transdifferentiation. Acute alveolar injury rapidly expands the AT2 stem cell pool by transiently inducing Wnt signaling activity in "bulk" AT2 cells, facilitating rapid epithelial repair. AT2 cell "stemness" is thus tightly regulated by access to Wnts, supplied by a specialized single-cell fibroblast niche during maintenance and by AT2 cells themselves during injury repair. Two non-AT2 "reserve" cell populations residing in the distal airways also contribute to alveolar repair, but only after widespread epithelial injury, when they rapidly proliferate, migrate, and differentiate into airway and alveolar lineages. Here, we review alveolar renewal and repair with a focus on the niches, rather than the stem cells, highlighting what is known about the cellular and molecular mechanisms by which they control stem cell activity in vivo.
Copyright © 2020 Cold Spring Harbor Laboratory Press; all rights reserved.
Figures

References
-
- Adamson IY, Bowden DH. 1974. The type 2 cell as progenitor of alveolar epithelial regeneration. A cytodynamic study in mice after exposure to oxygen. Lab Invest 30: 35–42. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
