Copper-catalyzed enantioselective conjugate addition of organometallic reagents to challenging Michael acceptors
- PMID: 32180841
- PMCID: PMC7059538
- DOI: 10.3762/bjoc.16.24
Copper-catalyzed enantioselective conjugate addition of organometallic reagents to challenging Michael acceptors
Abstract
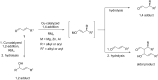
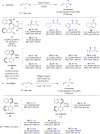
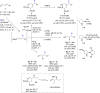
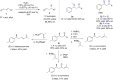
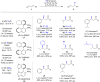
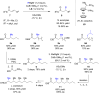
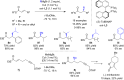
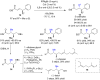
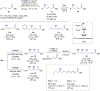
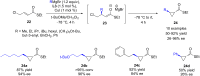
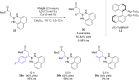
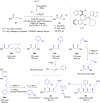
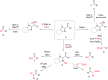
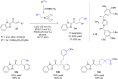
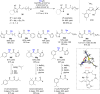
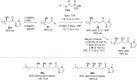
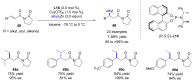
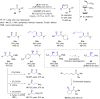
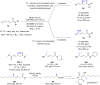
The copper-catalyzed enantioselective conjugate addition (ECA) of organometallic nucleophiles to electron-deficient alkenes (Michael acceptors) represents an efficient and attractive methodology for providing a wide range of relevant chiral molecules. In order to increase the attractiveness of this useful catalytic transformation, some Michael acceptors bearing challenging electron-deficient functions (i.e., aldehydes, thioesters, acylimidazoles, N-acyloxazolidinones, N-acylpyrrolidinones, amides, N-acylpyrroles) were recently investigated. Remarkably, only a few chiral copper-based catalytic systems have successfully achieved the conjugate addition of different organometallic reagents to these challenging Michael acceptors, with excellent regio- and enantioselectivity. Furthermore, thanks to their easy derivatization, the resulting chiral conjugated products could be converted into various natural products. The aim of this tutorial review is to summarize recent advances accomplished in this stimulating field.
Keywords: Michael acceptor; N-acyloxazolidinone; N-acylpyrrole; N-acylpyrrolidinone; acylimidazole; aldehyde; amide; copper catalysis; electron-deficient alkenes; enantioselective conjugate addition; thioester.
Copyright © 2020, Pichon et al.; licensee Beilstein-Institut.
Figures
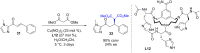


References
-
- Mauduit M, Baslé O, Clavier H, Crévisy C, Denicourt-Nowicki A. Metal-Catalyzed Asymmetric Nucleophilic Addition to Electron-Deficient Alkenes. In: Molander G A, Knochel P, editors. Comprehensive Organic Synthesis. 2nd ed. Vol. 4. Amsterdam, Netherlands: Elsevier; 2014. pp. 189–341. - DOI
-
- Alexakis A, Frutos J, Mangeney P. Tetrahedron: Asymmetry. 1993;4:2427–2430. doi: 10.1016/s0957-4166(00)82216-8. - DOI
Publication types
LinkOut - more resources
Full Text Sources
