Immune responses in liver and spleen against Plasmodium yoelii pre-erythrocytic stages in Swiss mice model
- PMID: 32181014
- PMCID: PMC7063113
- DOI: 10.1016/j.jare.2020.02.016
Immune responses in liver and spleen against Plasmodium yoelii pre-erythrocytic stages in Swiss mice model
Abstract
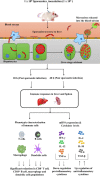
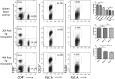
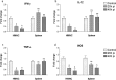
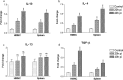
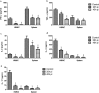
Though the immunity to malaria has been associated with cellular immune responses, the exact function of the phenotypic cell population is still unclear. This study investigated the host immune responses elicited during the pre-erythrocytic stage, post-Plasmodium yoelii sporozoite infection in Swiss mice model. For this purpose, we analyzed the dynamics of different subsets of immune cells population and cytokine levels in the hepatic mononuclear and splenic cells population during pre-erythrocytic liver-stage infection. We observed a significant reduction in the effectors immune cells population including CD8+ T cell, F4/80+ macrophage and in plasmacytoid dendritic cells (CD11c+ B220+). Interestingly, substantial down-regulation was also noted in pro-inflammatory cytokines (i.e. IFN-γ, TNF-α, IL-12, IL-2, IL-17 and iNOS), while, up-regulation of anti-inflammatory cytokines (i.e. IL-10, IL-4 and TGF-β) during asymptomatic pre-erythrocytic liver-stage infection. Collectively, this study demonstrated that during pre-erythrocytic development, Plasmodium yoelii sporozoite impaired the host activators of innate and adaptive immune responses by regulating the immune effector cells, gene expression and cytokines levels for the establishment of infection and subsequent development in the liver and spleen. The results in this study provided a better understanding of the events leading to malarial infection and will be helpful in supportive treatment and vaccine development strategy.
Keywords: Immune responses; Plasmodium yoelii; Pre-erythrocytic stage; Splenic cells; Swiss mice; T cells.
© 2020 THE AUTHORS. Published by Elsevier BV on behalf of Cairo University.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures






References
-
- WHO: World Health Organization. World Malaria Report Geneva; 2018. Available online at: http://www.who.int/malaria/publications/world-malaria-report-2018/en/. 2018.
-
- Soni A., Goyal M., Prakash K., Bhardwaj J., Siddiqui A.J., Puri S.K. Cloning, expression and functional characterization of heme detoxification protein (HDP) from the rodent malaria parasite Plasmodium vinckei. Gene. 2015;566:109–119. - PubMed
-
- Prakash K., Goyal M., Soni A., Siddiqui A.J., Bhardwaj J., Puri S.K. Molecular cloning and biochemical characterization of iron superoxide dismutase from the rodent malaria parasite Plasmodium vinckei. Parasitol Int. 2014;63:817–825. - PubMed
-
- Bhardwaj J., Siddiqui A.J., Goyal M., Prakash K., Soni A., Puri S.K. Repetitive live sporozoites inoculation under arteether chemoprophylaxis confers protection against subsequent sporozoite challenge in rodent malaria model. Acta Trop. 2016;158:130–138. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

