Dendritic Cell Maturation Regulates TSPAN7 Function in HIV-1 Transfer to CD4+ T Lymphocytes
- PMID: 32181159
- PMCID: PMC7059179
- DOI: 10.3389/fcimb.2020.00070
Dendritic Cell Maturation Regulates TSPAN7 Function in HIV-1 Transfer to CD4+ T Lymphocytes
Abstract
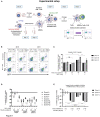
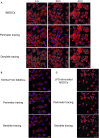
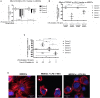
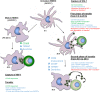
Dendritic cells (DCs) serve a key function in host defense, linking innate detection of microbes to activation of pathogen-specific adaptive immune responses. DCs express cell surface receptors for HIV-1 entry, but are relatively resistant to productive viral replication. They do, however, facilitate infection of co-cultured T-helper cells through a process referred to as trans-infection. We previously showed that tetraspanin 7 (TSPAN7), a transmembrane protein, is involved, through positive regulation of actin nucleation, in the transfer of HIV-1 from the dendrites of immature monocyte-derived DCs (iMDDCs) to activated CD4+ T lymphocytes. Various molecular mechanisms have been described regarding HIV-1 trans-infection and seem to depend on DC maturation status. We sought to investigate the crosstalk between DC maturation status, TSPAN7 expression and trans-infection. We followed trans-infection through co-culture of iMDDCs with CD4+ T lymphocytes, in the presence of CXCR4-tropic replicative-competent HIV-1 expressing GFP. T cell infection, DC maturation status and dendrite morphogenesis were assessed through time both by flow cytometry and confocal microscopy. Our previously described TSPAN7/actin nucleation-dependent mechanism of HIV-1 transfer appeared to be mostly observed during the first 20 h of co-culture experiments and to be independent of HIV replication. In the course of co-culture experiments, we observed a progressive maturation of MDDCs, correlated with a decrease in TSPAN7 expression, a drastic loss of dendrites and a change in the shape of DCs. A TSPAN7 and actin nucleation-independent mechanism of trans-infection, relying on HIV-1 replication, was then at play. We discovered that TSPAN7 expression is downregulated in response to different innate immune stimuli driving DC maturation, explaining the requirement for a TSPAN7/actin nucleation-independent mechanism of HIV transfer from mature MDDCs (mMDDCs) to T lymphocytes. As previously described, this mechanism relies on the capture of HIV-1 by the I-type lectin CD169/Siglec-1 on mMDDCs and the formation of a "big invaginated pocket" at the surface of DCs, both events being tightly regulated by DC maturation. Interestingly, in iMDDCs, although CD169/Siglec-1 can capture HIV-1, this capture does not lead to HIV-1 transfer to T lymphocytes.
Keywords: HIV-1; TSPAN7; actin nucleation; dendritic cell maturation; kinetic of transfer; trans-infection.
Copyright © 2020 Perot, García-Paredes, Luka and Ménager.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

