Cardiovascular Calcification in Chronic Kidney Disease-Therapeutic Opportunities
- PMID: 32183352
- PMCID: PMC7150985
- DOI: 10.3390/toxins12030181
Cardiovascular Calcification in Chronic Kidney Disease-Therapeutic Opportunities
Abstract
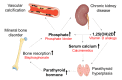
Patients with chronic kidney disease (CKD) are highly susceptible to cardiovascular (CV) complications, thus suffering from clinical manifestations such as heart failure and stroke. CV calcification greatly contributes to the increased CV risk in CKD patients. However, no clinically viable therapies towards treatment and prevention of CV calcification or early biomarkers have been approved to date, which is largely attributed to the asymptomatic progression of calcification and the dearth of high-resolution imaging techniques to detect early calcification prior to the 'point of no return'. Clearly, new intervention and management strategies are essential to reduce CV risk factors in CKD patients. In experimental rodent models, novel promising therapeutic interventions demonstrate decreased CKD-induced calcification and prevent CV complications. Potential diagnostic markers such as the serum T50 assay, which demonstrates an association of serum calcification propensity with all-cause mortality and CV death in CKD patients, have been developed. This review provides an overview of the latest observations and evaluates the potential of these new interventions in relation to CV calcification in CKD patients. To this end, potential therapeutics have been analyzed, and their properties compared via experimental rodent models, human clinical trials, and meta-analyses.
Keywords: cardiovascular disease; chronic kidney disease; experimental rodent models; vascular calcification.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Metra M., Zaca V., Parati G., Agostoni P., Bonadies M., Ciccone M., Cas A.D., Iacoviello M., Lagioia R., Lombardi C., et al. Cardiovascular and noncardiovascular comorbidities in patients with chronic heart failure. J. Cardiovasc. Med. 2011;12:76–84. doi: 10.2459/JCM.0b013e32834058d1. - DOI - PubMed
-
- Barreto F.C., Barreto D.V., Liabeuf S., Meert N., Glorieux G., Temmar M., Choukroun G., Vanholder R., Massy Z.A. Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin. J. Am. Soc. Nephrol. 2009;4:1551–1558. doi: 10.2215/CJN.03980609. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

