Implementation of the patient version of the evidence-based (S3) guideline for psychosocial interventions for patients with severe mental illness (IMPPETUS): study protocol for a cluster randomised controlled trial
- PMID: 32183897
- PMCID: PMC7079434
- DOI: 10.1186/s13063-020-4200-z
Implementation of the patient version of the evidence-based (S3) guideline for psychosocial interventions for patients with severe mental illness (IMPPETUS): study protocol for a cluster randomised controlled trial
Abstract
Background: The German guideline on psychosocial interventions for people with severe mental disorders recommends a broad spectrum of evidence-based treatments. Structured implementation of the associated patient version of the guideline is missing to date. The study aims to assess whether structured implementation of a patient guideline improves the empowerment of patients with severe mental disorders, as well as knowledge, attitudes and experiences regarding psychosocial interventions, service use, treatment satisfaction, treatment needs, quality of life and burden of care.
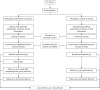
Methods: The study is a multicentre, cluster-randomised, controlled study with two parallel groups. Inpatients and day hospital patients (all sexes; 18-65 years) with severe mental disorders will be included. Additionally, relatives of patients with mental disorders (all sexes; ≥ 18 years) will be included. In the experimental group, the patient guideline will be implemented using a multimodal strategy. Participants in the control group will receive treatment as usual but will be made aware of the patient guideline. The primary outcome is the change of empowerment, assessed by using the 'empowerment in the process of psychiatric treatment of patients with affective and schizophrenia disorders' (EPAS) scale. In addition, knowledge, attitudes and experiences regarding psychosocial interventions will be assessed as secondary outcomes, as well as service use, satisfaction with care, patient need and quality of life and participation and social inclusion. For relatives, the perceived burden of care also will be recorded. Results will be analysed using hierarchical linear models. For the health economic evaluation, the incremental cost-utility ratios will be computed using the differences in total costs of illness and the differences in quality-adjusted life years (QALY) between study groups.
Discussion: The study will be the first to assess the effects of a structured implementation of the patient version of a psychiatric treatment guideline. The study has some limitations regarding the transferability of the results to other patients and other regions. Furthermore, problems with the recruitment of patients and relatives and with the implementation of intervention could occur during the study.
Trial registration: The study is registered in the German Clinical Trials Register (DRKS) and the WHO International Clinical Trials Registry Platform (ICTRP) under registration number DRKS00017577 (Date of registration: 23 October 2019.
Keywords: Guideline, Implementation, Patient version, Cluster randomised controlled trial.
Conflict of interest statement
The authors declare that they have no competing interests.
Similar articles
-
Systematic reviews of the effectiveness of day care for people with severe mental disorders: (1) acute day hospital versus admission; (2) vocational rehabilitation; (3) day hospital versus outpatient care.Health Technol Assess. 2001;5(21):1-75. doi: 10.3310/hta5210. Health Technol Assess. 2001. PMID: 11532238 Review.
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
Effectiveness and cost-effectiveness of a community-based mental health care programme (GBV) for people with severe mental illness in Germany: study protocol for a randomised controlled trial.Trials. 2020 Jun 30;21(1):598. doi: 10.1186/s13063-020-04492-y. Trials. 2020. PMID: 32605585 Free PMC article.
-
Peer support for people with severe mental illness versus usual care in high-, middle- and low-income countries: study protocol for a pragmatic, multicentre, randomised controlled trial (UPSIDES-RCT).Trials. 2020 May 1;21(1):371. doi: 10.1186/s13063-020-4177-7. Trials. 2020. PMID: 32357903 Free PMC article.
-
Behavioural modification interventions for medically unexplained symptoms in primary care: systematic reviews and economic evaluation.Health Technol Assess. 2020 Sep;24(46):1-490. doi: 10.3310/hta24460. Health Technol Assess. 2020. PMID: 32975190 Free PMC article.
Cited by
-
The role of migration in mental healthcare: treatment satisfaction and utilization.BMC Psychiatry. 2022 Feb 15;22(1):116. doi: 10.1186/s12888-022-03722-8. BMC Psychiatry. 2022. PMID: 35168572 Free PMC article.
References
-
- Gühne U, Weinmann S, Riedel-Heller SG, Becker T. S3-Leitlinie Psychosoziale Therapien bei schweren psychischen Erkrankungen: S3-Praxisleitlinien in Psychiatrie und Psychotherapie [German]. Herausgebende Fachgesellschaft: DGPPN. 2. Berlin: Springer; 2019.
-
- Gühne U, Fricke R, Schliebener G, Becker T, Riedel-Heller SG. Psychosoziale Therapien bei schweren psychischen Erkrankungen - Patientenleitlinie für Betroffene und Angehörige [German] 2. Berlin, Heidelberg: Springer; 2019.
-
- Kösters M, Staudigl L, Picca A-C, Schmauß M, Becker T, Weinmann S. Qualitätsindikatoren für die Behandlung von Menschen mit Schizophrenie – Ergebnisse einer Anwendungsstudie [German] Psychiatr Prax. 2017;44:163–171. - PubMed
-
- Bramesfeld A, Schäfer I, Stengler K, Schomerus G. Impulse für die Versorgungsforschung: Was folgt auf die DGPPN S3-Leitlinie zu psychosozialen Therapien? [German] Psychiatr Prax. 2014;41:65–67. - PubMed
-
- Girlanda F, Fiedler I, Becker T, Barbui C, Koesters M. The evidence-practice gap in specialist mental healthcare: systematic review and meta-analysis of guideline implementation studies. Br J Psychiatry. 2017;210:24–30. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical