The Response of Tissue Mast Cells to TLR3 Ligand Poly(I:C) Treatment
- PMID: 32185237
- PMCID: PMC7060451
- DOI: 10.1155/2020/2140694
The Response of Tissue Mast Cells to TLR3 Ligand Poly(I:C) Treatment
Abstract
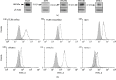
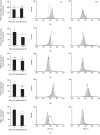
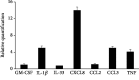
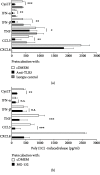
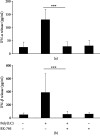
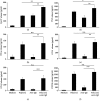
Mast cells (MCs) are found mainly at the anatomical sites exposed to the external environment; thus, they are localized close to blood vessels, lymphatic vessels, and a multitude of immune cells. Moreover, those cells can recognize invading pathogens through a range of surface molecules known as pathogen recognition receptors (PRRs), mainly Toll-like receptors (TLRs). MCs are extensively engaged in the control and clearance of bacterial infections, but much less is known about their contribution to antiviral host response as well as pathomechanisms of virus-induced diseases. In the study, we employed in vivo differentiated mature tissue mast cells freshly isolated from rat peritoneal cavity. Here, we demonstrated that rat peritoneal mast cells (rPMCs) express viral dsRNA-specific TLR3 molecule (intracellularly and on the cell surface) as well as other proteins associated with cellular antiviral response: IRF3, type I and II IFN receptors, and MHC I. We found that exposure of rPMCs to viral dsRNA mimic, i.e., poly(I:C), induced transient upregulation of surface TLR3 (while temporarily decreased TLR3 intracellular expression), type II IFN receptor, and MHC I. TLR3 ligand-stimulated rPMCs did not degranulate but generated and/or released type I IFNs (IFN-α and IFNβ) as well as proinflammatory lipid mediators (cysLTs), cytokines (TNF, IL-1β), and chemokines (CCL3, CXCL8). We documented that rPMC priming with poly(I:C) did not affect FcεRI-dependent degranulation. However, their costimulation with TLR3 agonist and anti-IgE led to a significant increase in cysLT and TNF secretion. Our findings confirm that MCs may serve as active participants in the antiviral immune response. Presented data on modulated FcεRI-mediated MC secretion of mediators upon poly(I:C) treatment suggests that dsRNA-type virus infection could influence the severity of allergic reactions.
Copyright © 2020 Piotr Witczak et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

