Benzothiazolyl Ureas are Low Micromolar and Uncompetitive Inhibitors of 17β-HSD10 with Implications to Alzheimer's Disease Treatment
- PMID: 32192199
- PMCID: PMC7139388
- DOI: 10.3390/ijms21062059
Benzothiazolyl Ureas are Low Micromolar and Uncompetitive Inhibitors of 17β-HSD10 with Implications to Alzheimer's Disease Treatment
Abstract
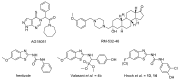
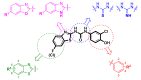
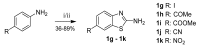
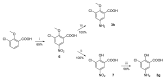
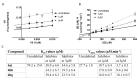
Human 17β-hydroxysteroid dehydrogenase type 10 is a multifunctional protein involved in many enzymatic and structural processes within mitochondria. This enzyme was suggested to be involved in several neurological diseases, e.g., mental retardation, Parkinson's disease, or Alzheimer's disease, in which it was shown to interact with the amyloid-beta peptide. We prepared approximately 60 new compounds based on a benzothiazolyl scaffold and evaluated their inhibitory ability and mechanism of action. The most potent inhibitors contained 3-chloro and 4-hydroxy substitution on the phenyl ring moiety, a small substituent at position 6 on the benzothiazole moiety, and the two moieties were connected via a urea linker (4at, 4bb, and 4bg). These compounds exhibited IC50 values of 1-2 μM and showed an uncompetitive mechanism of action with respect to the substrate, acetoacetyl-CoA. These uncompetitive benzothiazolyl inhibitors of 17β-hydroxysteroid dehydrogenase type 10 are promising compounds for potential drugs for neurodegenerative diseases that warrant further research and development.
Keywords: 17β-hydroxysteroid dehydrogenase type 10; ABAD; Alzheimer’s disease; benzothiazole; inhibitor; neurodegeneration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
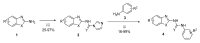

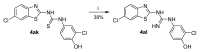
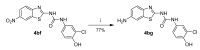
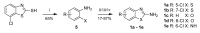
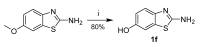
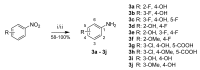
References
-
- He X.Y., Merz G., Mehta P., Schulz H., Yang S.Y. Human brain short chain L-3-hydroxyacyl coenzyme A dehydrogenase is a single-domain multifunctional enzyme. Characterization of a novel 17beta-hydroxysteroid dehydrogenase. J. Biol. Chem. 1999;274:15014–15019. doi: 10.1074/jbc.274.21.15014. - DOI - PubMed
-
- Zschocke J., Ruiter J.P., Brand J., Lindner M., Hoffmann G.F., Wanders R.J., Mayatepek E. Progressive infantile neurodegeneration caused by 2-methyl-3-hydroxybutyryl-CoA dehydrogenase deficiency: A novel inborn error of branched-chain fatty acid and isoleucine metabolism. Pediatr. Res. 2000;48:852–855. doi: 10.1203/00006450-200012000-00025. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

