Hepatic Transcript Profiles of Cytochrome P450 Genes Predict Sex Differences in Drug Metabolism
- PMID: 32193355
- PMCID: PMC7250365
- DOI: 10.1124/dmd.119.089367
Hepatic Transcript Profiles of Cytochrome P450 Genes Predict Sex Differences in Drug Metabolism
Abstract
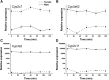
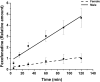
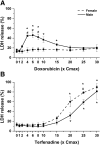
Safety assessments of new drug candidates are an important part of the drug development and approval process. Often, possible sex-associated susceptibilities are not adequately addressed, and better assessment tools are needed. We hypothesized that hepatic transcript profiles of cytochrome P450 (P450) enzymes can be used to predict sex-associated differences in drug metabolism and possible adverse events. Comprehensive hepatic transcript profiles were generated for F344 rats of both sexes at nine ages, from 2 weeks (preweaning) to 104 weeks (elderly). Large differences in the transcript profiles of 29 drug metabolizing enzymes and transporters were found between adult males and females (8-52 weeks). Using the PharmaPendium data base, 41 drugs were found to be metabolized by one or two P450 enzymes encoded by sexually dimorphic mRNAs and thus were candidates for evaluation of possible sexually dimorphic metabolism and/or toxicities. Suspension cultures of primary hepatocytes from three male and three female adult rats (10-13 weeks old) were used to evaluate the metabolism of 11 drugs predicted to have sexually dimorphic metabolism. The pharmacokinetics of the drug or its metabolite was analyzed by liquid chromatography/tandem mass spectrometry using multiple reaction monitoring. Of those drugs with adequate metabolism, the predicted significant sex-different metabolism was found for six of seven drugs, with half-lives 37%-400% longer in female hepatocytes than in male hepatocytes. Thus, in this rat model, transcript profiles may allow identification of potential sex-related differences in drug metabolism. SIGNIFICANCE STATEMENT: The present study showed that sex-different expression of genes coding for drug metabolizing enzymes, specifically cytochrome P450s, could be used to predict sex-different drug metabolism and, thus, provide a new tool for protecting susceptible subpopulations from possible adverse drug events.
U.S. Government work not protected by U.S. copyright.
Figures




References
-
- Arrowsmith J. (2011) Trial watch: phase III and submission failures: 2007-2010. Nat Rev Drug Discov 10:87. - PubMed
-
- Attar M, Dong D, Ling KH, Tang-Liu DD. (2003) Cytochrome P450 2C8 and flavin-containing monooxygenases are involved in the metabolism of tazarotenic acid in humans. Drug Metab Dispos 31:476–481. - PubMed
-
- Cho S, Lau SW, Tandon V, Kumi K, Pfuma E, Abernethy DR. (2011) Geriatric drug evaluation: where are we now and where should we be in the future? Arch Intern Med 171:937–940. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

