Parental repeat length instability in myotonic dystrophy type 1 pre- and protomutations
- PMID: 32203199
- PMCID: PMC7316980
- DOI: 10.1038/s41431-020-0601-4
Parental repeat length instability in myotonic dystrophy type 1 pre- and protomutations
Abstract
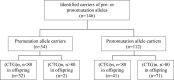
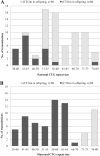
Myotonic dystrophy type 1 (DM1) is caused by a CTG trinucleotide repeat expansion on chromosome 19q13.3. While DM1 premutation (36-50 repeats) and protomutation (51-80 repeats) allele carriers are mostly asymptomatic, offspring is at risk of inheriting expanded, symptom-associated, (CTG)n repeats of n > 80. In this study we aimed to evaluate the intergenerational instability of DM1 pre- and protomutation alleles, focussing on the influence of parental gender. One hundred and forty-six parent-child pairs (34 parental premutations, 112 protomutations) were retrospectively selected from the DM1 patient cohort of the Maastricht University Medical Center+. CTG repeat size of parents and children was determined by (triplet-primed) PCR followed by fragment length analysis and Southern blot analysis. Fifty-eight out of eighty-one (71.6%) paternal transmissions led to a (CTG)n repeat of n > 80 in offspring, compared with 15 out of 65 (23.1%) maternal transmissions (p < 0.001). Repeat length instability occurred for paternal (CTG)n repeats of n ≥ 45, while maternal instability did not occur until (CTG)n repeats reached a length of n ≥ 71. Transmission of premutations caused (CTG)n repeats of n > 80 in offspring only when paternally transmitted (two cases), while protomutations caused (CTG)n repeats of n > 80 in offspring in 71 cases, of which 56 (78.9%) were paternally transmitted. In conclusion, our data show that paternally transmitted pre- and protomutations were more unstable than maternally transmitted pre- and protomutations. For genetic counseling, this implies that males with a small DMPK mutation have a higher risk of symptomatic offspring compared with females. Consequently, we suggest addressing sex-dependent factors in genetic counseling of small-sized CTG repeat carriers.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

Similar articles
-
Instability of a premutation allele in homozygous patients with myotonic dystrophy type 1.Ann Neurol. 2002 Oct;52(4):435-41. doi: 10.1002/ana.10304. Ann Neurol. 2002. PMID: 12325072
-
Frequency of intergenerational contractions of the CTG repeats in myotonic dystrophy.Genet Epidemiol. 1996;13(5):483-7. doi: 10.1002/(SICI)1098-2272(1996)13:5<483::AID-GEPI4>3.0.CO;2-3. Genet Epidemiol. 1996. PMID: 8905394
-
Anticipation in myotonic dystrophy type 1 parents with small CTG expansions.Am J Med Genet A. 2015 Apr;167A(4):708-14. doi: 10.1002/ajmg.a.36950. Epub 2015 Feb 25. Am J Med Genet A. 2015. PMID: 25712547
-
Overview of the Complex Relationship between Epigenetics Markers, CTG Repeat Instability and Symptoms in Myotonic Dystrophy Type 1.Int J Mol Sci. 2022 Mar 23;23(7):3477. doi: 10.3390/ijms23073477. Int J Mol Sci. 2022. PMID: 35408837 Free PMC article. Review.
-
Molecular genetics and genetic testing in myotonic dystrophy type 1.Biomed Res Int. 2013;2013:391821. doi: 10.1155/2013/391821. Epub 2013 Mar 18. Biomed Res Int. 2013. PMID: 23586035 Free PMC article. Review.
Cited by
-
Interrupted CTG repeats in the 37-43 units size range in the 3'UTR of DMPK are common alleles.Eur J Hum Genet. 2025 Jul 8. doi: 10.1038/s41431-025-01907-9. Online ahead of print. Eur J Hum Genet. 2025. PMID: 40624398
-
Somatic and intergenerational G4C2 hexanucleotide repeat instability in a human C9orf72 knock-in mouse model.Nucleic Acids Res. 2024 Jun 10;52(10):5732-5755. doi: 10.1093/nar/gkae250. Nucleic Acids Res. 2024. PMID: 38597682 Free PMC article.
-
Intergenerational Influence of Gender and the DM1 Phenotype of the Transmitting Parent in Korean Myotonic Dystrophy Type 1.Genes (Basel). 2022 Aug 17;13(8):1465. doi: 10.3390/genes13081465. Genes (Basel). 2022. PMID: 36011377 Free PMC article.
-
Multisystem Symptoms in Myotonic Dystrophy Type 1: A Management and Therapeutic Perspective.Int J Mol Sci. 2025 Jun 2;26(11):5350. doi: 10.3390/ijms26115350. Int J Mol Sci. 2025. PMID: 40508159 Free PMC article. Review.
-
Optical Genome Mapping Enables Detection and Accurate Sizing of RFC1 Repeat Expansions.Biomolecules. 2023 Oct 19;13(10):1546. doi: 10.3390/biom13101546. Biomolecules. 2023. PMID: 37892228 Free PMC article.
References
-
- Brook JD, McCurrach ME, Harley HG, Buckler AJ, Church D, Aburatani H, et al. Molecular basis of myotonic dystrophy: expansion of a trinucleotide (CTG) repeat at the 3’ end of a transcript encoding a protein kinase family member. Cell. 1992;69:385. - PubMed
-
- Gatchel JR, Zoghbi HY. Diseases of unstable repeat expansion: mechanisms and common principles. Nat Rev Genet. 2005;6:743–55. - PubMed
-
- Harper PS. Myotonic dystrophy present management, future therapy. Oxford: Oxford University Press; 2004. - PubMed
-
- Meola G, Cardani R. Myotonic dystrophies: an update on clinical aspects, genetic, pathology, and molecular pathomechanisms. Biochimica et Biophysica Acta. 2015;1852:594–606. - PubMed
-
- Imbert G, Kretz C, Johnson K, Mandel JL. Origin of the expansion mutation in myotonic dystrophy. Nat Genet. 1993;4:72–6. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

