Immunization Against Oxidized Elastin Exacerbates Structural and Functional Damage in Mouse Model of Smoke-Induced Ocular Injury
- PMID: 32207814
- PMCID: PMC7401451
- DOI: 10.1167/iovs.61.3.45
Immunization Against Oxidized Elastin Exacerbates Structural and Functional Damage in Mouse Model of Smoke-Induced Ocular Injury
Erratum in
-
Erratum in: Immunization Against Oxidized Elastin Exacerbates Structural and Functional Damage in Mouse Model of Smoke-Induced Ocular Injury.Invest Ophthalmol Vis Sci. 2021 Apr 1;62(4):13. doi: 10.1167/iovs.62.4.13. Invest Ophthalmol Vis Sci. 2021. PMID: 33844827 Free PMC article. No abstract available.
Abstract
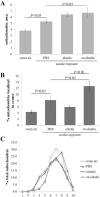
Purpose: Age-related macular degeneration (AMD) is the leading cause of blindness in Western populations. While an overactive complement system has been linked to pathogenesis, mechanisms contributing to its activation are largely unknown. In aged and AMD eyes, loss of the elastin layer (EL) of Bruch's membrane (BrM) has been reported. Elastin antibodies are elevated in patients with AMD, the pathogenic significance of which is unclear. Here we assess the role of elastin antibodies using a mouse model of smoke-induced ocular pathology (SIOP), which similarly demonstrates EL loss.
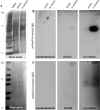
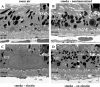
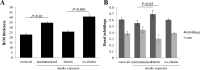
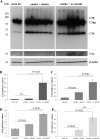
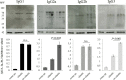
Methods: C57BL/6J mice were immunized with elastin or elastin peptide oxidatively modified by cigarette smoke (ox-elastin). Mice were then exposed to cigarette smoke or air for 6 months. Visual function was assessed by optokinetic response, retinal morphology by spectral-domain optical coherence tomography and electron microscopy, and complement activation and antibody deposition by Western blot.
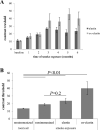
Results: Ox-elastin IgG and IgM antibodies were elevated in ox-elastin immunized mice following 6 months of smoke, whereas elastin immunization had a smaller effect. Ox-elastin immunization exacerbated smoke-induced vision loss, with thicker BrM and more damaged retinal pigment epithelium (RPE) mitochondria compared with mice immunized with elastin or nonimmunized controls. These changes were correlated with increased levels of IgM, IgG2, IgG3, and complement activation products in RPE/choroid.
Conclusions: These data demonstrate that SIOP mice generate elastin-specific antibodies and that immunization with ox-elastin exacerbates ocular pathology. Elastin antibodies represented complement fixing isotypes that, together with the increased presence of complement activation seen in immunized mice, suggest that elastin antibodies exert pathogenic effects through mediating complement activation.
Conflict of interest statement
Disclosure:
Figures



References
-
- Brown MM, Brown GC, Stein JD, Roth Z, Campanella J, Beauchamp GR. Age-related macular degeneration: economic burden and value-based medicine analysis. Can J Ophthalmol. 2005; 40: 277–287. - PubMed
-
- Johnson LV, Leitner WP, Staples MK, Anderson DH. Complement activation and inflammatory processes in Drusen formation and age related macular degeneration. Exp Eye Res. 2001; 73: 887–896. - PubMed
-
- Umeda S, Suzuki MT, Okamoto H, et al. .. Molecular composition of drusen and possible involvement of anti-retinal autoimmunity in two different forms of macular degeneration in cynomolgus monkey (Macaca fascicularis). Faseb J. 2005; 19: 1683–1685. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

