Detection of amastigotes and histopathological alterations in the thymus of Leishmania infantum-infected dogs
- PMID: 32207879
- PMCID: PMC7212199
- DOI: 10.1002/iid3.285
Detection of amastigotes and histopathological alterations in the thymus of Leishmania infantum-infected dogs
Abstract
Introduction: In canine visceral leishmaniasis (CVL), lymphopenia, and the disorganization of lymphoid organs such as spleen and lymph nodes have been demonstrated. However, the involvement of thymus in CVL has not been evaluated so far. Herein, we investigated whether the thymus can be colonized by Leishmania infantum in naturally infected dogs.
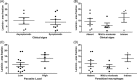
Methods: Thymus were obtained from 16 of 58 dogs and samples of this organ were submitted to immunohistochemistry for laminin and fibronectin detection, histopathology, in situ hybridization and polymerase chain reaction (PCR) targeting the gene ITS-1 for Leishmania and sequenced. Samples of spleen, skin and popliteal lymph nodes were collected and submitted to immunohistochemistry and parasitological culture followed by multilocus enzyme electrophoresis.
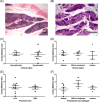
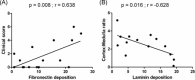
Results: L. infantum was identified in all dogs. DNA and amastigote forms of Leishmania were detected in the thymus from 16 dogs by PCR and in eight by immunohistochemistry. Besides thymus, parasites were detected in spleen, lymph nodes, and skin. A granulomatous or pyogranulomatous thymitis was observed in eight dogs associated to intact amastigotes forms of this parasite. Fibronectin deposition in thymus was higher in dogs with more clinical signs.
Conclusions: These results demonstrate that the thymus of dogs can be parasitized by L. infantum, which may generate inflammatory reactions leading to alterations in thymic microarchitecture.
Keywords: Leishmania infantum; canine visceral leishmaniasis; clinical signs; extracellular matrix; histopathology; parasite load; thymus.
© 2020 The Authors. Immunity, Inflammation and Disease published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare that there are no conflict of interests.
Figures


References
-
- Borja LS, Sousa OMF, Solcà MS, et al. Parasite load in the blood and skin of dogs naturally infected by Leishmania infantum is correlated with their capacity to infect sand fly vectors. Vet Parasitol. 2016;229:110‐117. - PubMed
-
- Koutinas AF, Koutinas CK. Pathologic mechanisms underlying the clinical findings in canine leishmaniasis due to Leishmania infantum/chagasi. Vet Pathol. 2014;51:527‐538. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

