Interferon-complement loop in transplant-associated thrombotic microangiopathy
- PMID: 32208488
- PMCID: PMC7094010
- DOI: 10.1182/bloodadvances.2020001515
Interferon-complement loop in transplant-associated thrombotic microangiopathy
Abstract
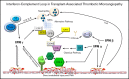
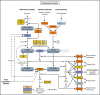
Transplant-associated thrombotic microangiopathy (TA-TMA) is an important cause of morbidity and mortality after hematopoietic stem cell transplantation (HSCT). The complement inhibitor eculizumab improves TA-TMA, but not all patients respond to therapy, prompting a search for additional targetable pathways of endothelial injury. TA-TMA is relatively common after HSCT and can serve as a model to study mechanisms of tissue injury in other thrombotic microangiopathies. In this work, we performed transcriptome analyses of peripheral blood mononuclear cells collected before HSCT, at onset of TA-TMA, and after resolution of TA-TMA in children with and without TA-TMA after HSCT. We observed significant upregulation of the classical, alternative, and lectin complement pathways during active TA-TMA. Essentially all upregulated genes and pathways returned to baseline expression levels at resolution of TA-TMA after eculizumab therapy, supporting the clinical practice of discontinuing complement blockade after resolution of TA-TMA. Further analysis of the global transcriptional regulatory network showed a notable interferon signature associated with TA-TMA with increased STAT1 and STAT2 signaling that resolved after complement blockade. In summary, we observed activation of multiple complement pathways in TA-TMA, in contrast to atypical hemolytic uremic syndrome (aHUS), where complement activation occurs largely via the alternative pathway. Our data also suggest a key relationship between increased interferon signaling, complement activation, and TA-TMA. We propose a model of an "interferon-complement loop" that can perpetuate endothelial injury and thrombotic microangiopathy. These findings open opportunities to study novel complement blockers and combined anti-complement and anti-interferon therapies in patients with TA-TMA and other microangiopathies like aHUS and lupus-associated TMAs.
© 2020 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: S.J. and S.M.D. have National Institutes of Health–funded research for TA-TMA and received free drug for study subjects from Alexion, unrelated to this work. S.J. received travel support and honoraria for presentation at the scientific meeting from Omeros and consultancy fees from Arcus Medica and Magnolia Innovations, unrelated to this work. M.D. received consultancy fees from Novartis and research funding and travel support from Prolacta, all unrelated to this work. B.L.L. has received consulting fees from Jazz Pharmaceuticals, unrelated to this work. The remaining authors have no disclosures to report.
Figures



Similar articles
-
Complement blockade for TA-TMA: lessons learned from a large pediatric cohort treated with eculizumab.Blood. 2020 Mar 26;135(13):1049-1057. doi: 10.1182/blood.2019004218. Blood. 2020. PMID: 31932840 Free PMC article.
-
Terminal Complement Blockade after Hematopoietic Stem Cell Transplantation Is Safe without Meningococcal Vaccination.Biol Blood Marrow Transplant. 2016 Jul;22(7):1337-1340. doi: 10.1016/j.bbmt.2016.03.032. Epub 2016 Apr 7. Biol Blood Marrow Transplant. 2016. PMID: 27060440 Free PMC article.
-
Eculizumab therapy in children with severe hematopoietic stem cell transplantation-associated thrombotic microangiopathy.Biol Blood Marrow Transplant. 2014 Apr;20(4):518-25. doi: 10.1016/j.bbmt.2013.12.565. Epub 2013 Dec 25. Biol Blood Marrow Transplant. 2014. PMID: 24370861 Free PMC article.
-
Transplant-Associated Thrombotic Microangiopathy in the Context of Allogenic Hematopoietic Stem Cell Transplantation: Where We Stand.Int J Mol Sci. 2023 Jan 6;24(2):1159. doi: 10.3390/ijms24021159. Int J Mol Sci. 2023. PMID: 36674666 Free PMC article. Review.
-
Efficacy and Safety of Eculizumab in the Treatment of Transplant-Associated Thrombotic Microangiopathy: A Systematic Review and Meta-Analysis.Front Immunol. 2021 Jan 20;11:564647. doi: 10.3389/fimmu.2020.564647. eCollection 2020. Front Immunol. 2021. PMID: 33552043 Free PMC article.
Cited by
-
Evolution of SARS-CoV-2 Variants: Implications on Immune Escape, Vaccination, Therapeutic and Diagnostic Strategies.Viruses. 2023 Apr 10;15(4):944. doi: 10.3390/v15040944. Viruses. 2023. PMID: 37112923 Free PMC article. Review.
-
Role of Defective Interfering Particles in Complement-Mediated Lysis of Parainfluenza Virus-Infected Cells.Viruses. 2025 Mar 28;17(4):488. doi: 10.3390/v17040488. Viruses. 2025. PMID: 40284931 Free PMC article.
-
Genetic Susceptibility in Sinusoidal Obstruction Syndrome/Veno-Occlusive Disease: A Case-Control Study.Int J Mol Sci. 2025 Jul 12;26(14):6712. doi: 10.3390/ijms26146712. Int J Mol Sci. 2025. PMID: 40724958 Free PMC article.
-
Thinking Beyond HLH: Clinical Features of Patients with Concurrent Presentation of Hemophagocytic Lymphohistiocytosis and Thrombotic Microangiopathy.J Clin Immunol. 2020 Jul;40(5):699-707. doi: 10.1007/s10875-020-00789-4. Epub 2020 May 23. J Clin Immunol. 2020. PMID: 32447592 Free PMC article.
-
Hematopoietic stem cell transplantation-associated thrombotic microangiopathy and the role of advanced practice providers and pharmacists.Bone Marrow Transplant. 2023 Jun;58(6):625-634. doi: 10.1038/s41409-023-01951-3. Epub 2023 Apr 14. Bone Marrow Transplant. 2023. PMID: 37059738 Free PMC article. Review.
References
-
- Cho BS, Yahng SA, Lee SE, et al. . Validation of recently proposed consensus criteria for thrombotic microangiopathy after allogeneic hematopoietic stem-cell transplantation. Transplantation. 2010;90(8):918-926. - PubMed
-
- Chima RS, Abulebda K, Jodele S. Advances in critical care of the pediatric hematopoietic stem cell transplant patient. Pediatr Clin North Am. 2013;60(3):689-707. - PubMed
-
- Fernández-García M, Gonzalez-Vicent M, Mastro-Martinez I, Serrano A, Diaz MA. Intensive care unit admissions among children after hematopoietic stem cell transplantation: incidence, outcome, and prognostic factors. J Pediatr Hematol Oncol. 2015;37(7):529-535. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

