Disorders of Sex Development-Novel Regulators, Impacts on Fertility, and Options for Fertility Preservation
- PMID: 32224856
- PMCID: PMC7178030
- DOI: 10.3390/ijms21072282
Disorders of Sex Development-Novel Regulators, Impacts on Fertility, and Options for Fertility Preservation
Abstract
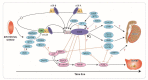
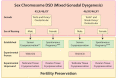
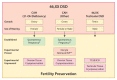
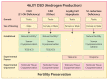
Disorders (or differences) of sex development (DSD) are a heterogeneous group of congenital conditions with variations in chromosomal, gonadal, or anatomical sex. Impaired gonadal development is central to the pathogenesis of the majority of DSDs and therefore a clear understanding of gonadal development is essential to comprehend the impacts of these disorders on the individual, including impacts on future fertility. Gonadal development was traditionally considered to involve a primary 'male' pathway leading to testicular development as a result of expression of a small number of key testis-determining genes. However, it is increasingly recognized that there are several gene networks involved in the development of the bipotential gonad towards either a testicular or ovarian fate. This includes genes that act antagonistically to regulate gonadal development. This review will highlight some of the novel regulators of gonadal development and how the identification of these has enhanced understanding of gonadal development and the pathogenesis of DSD. We will also describe the impact of DSDs on fertility and options for fertility preservation in this context.
Keywords: disorder of sex development; fertility; fertility preservation; gonads; ovary; sex determination; testis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Bashamboo A., Donohoue P.A., Vilain E., Rojo S., Calvel P., Seneviratne S.N., Buonocore F., Barseghyan H., Bingham N., Rosenfeld J.A., et al. A recurrent p.Arg92Trp variant in steroidogenic factor-1 (NR5A1) can act as a molecular switch in human sex development. Hum. Mol. Genet. 2016;25:3446–3453. doi: 10.1093/hmg/ddw186. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

