β3-Adrenoceptor redistribution impairs NO/cGMP/PDE2 signalling in failing cardiomyocytes
- PMID: 32228862
- PMCID: PMC7138611
- DOI: 10.7554/eLife.52221
β3-Adrenoceptor redistribution impairs NO/cGMP/PDE2 signalling in failing cardiomyocytes
Abstract
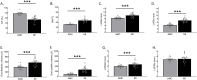
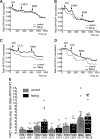
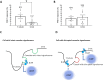
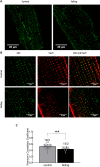
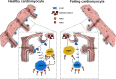
Cardiomyocyte β3-adrenoceptors (β3-ARs) coupled to soluble guanylyl cyclase (sGC)-dependent production of the second messenger 3',5'-cyclic guanosine monophosphate (cGMP) have been shown to protect from heart failure. However, the exact localization of these receptors to fine membrane structures and subcellular compartmentation of β3-AR/cGMP signals underpinning this protection in health and disease remain elusive. Here, we used a Förster Resonance Energy Transfer (FRET)-based cGMP biosensor combined with scanning ion conductance microscopy (SICM) to show that functional β3-ARs are mostly confined to the T-tubules of healthy rat cardiomyocytes. Heart failure, induced via myocardial infarction, causes a decrease of the cGMP levels generated by these receptors and a change of subcellular cGMP compartmentation. Furthermore, attenuated cGMP signals led to impaired phosphodiesterase two dependent negative cGMP-to-cAMP cross-talk. In conclusion, topographic and functional reorganization of the β3-AR/cGMP signalosome happens in heart failure and should be considered when designing new therapies acting via this receptor.
Keywords: cGMP; cardiomyocytes; cell biology; heart; rat; second messenger; signalling.
© 2020, Schobesberger et al.
Conflict of interest statement
SS, PW, CP, JS, CM, AF, SH, JB, VN, JG No competing interests declared
Figures




References
-
- Amour J, Loyer X, Le Guen M, Mabrouk N, David JS, Camors E, Carusio N, Vivien B, Andriantsitohaina R, Heymes C, Riou B. Altered contractile response due to increased beta3-adrenoceptor stimulation in diabetic cardiomyopathy: the role of nitric oxide synthase 1-derived nitric oxide. Anesthesiology. 2007;107:452–460. doi: 10.1097/01.anes.0000278909.40408.24. - DOI - PubMed
-
- Belge C, Hammond J, Dubois-Deruy E, Manoury B, Hamelet J, Beauloye C, Markl A, Pouleur AC, Bertrand L, Esfahani H, Jnaoui K, Götz KR, Nikolaev VO, Vanderper A, Herijgers P, Lobysheva I, Iaccarino G, Hilfiker-Kleiner D, Tavernier G, Langin D, Dessy C, Balligand JL. Enhanced expression of β3-adrenoceptors in cardiac myocytes attenuates neurohormone-induced hypertrophic remodeling through nitric oxide synthase. Circulation. 2014;129:451–462. doi: 10.1161/CIRCULATIONAHA.113.004940. - DOI - PubMed
-
- Cawley SM, Kolodziej S, Ichinose F, Brouckaert P, Buys ES, Bloch KD. sGC{alpha}1 mediates the negative inotropic effects of NO in cardiac myocytes independent of changes in calcium handling. American Journal of Physiology. Heart and Circulatory Physiology. 2011;301:H157–H163. doi: 10.1152/ajpheart.01273.2010. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

