Nanocellulose-Based Patches Loaded with Hyaluronic Acid and Diclofenac towards Aphthous Stomatitis Treatment
- PMID: 32231070
- PMCID: PMC7221765
- DOI: 10.3390/nano10040628
Nanocellulose-Based Patches Loaded with Hyaluronic Acid and Diclofenac towards Aphthous Stomatitis Treatment
Abstract
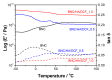
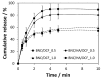
Nanostructured patches composed of bacterial nanocellulose (BNC), hyaluronic acid (HA) and diclofenac (DCF) were developed, envisioning the treatment of aphthous stomatitis. Freestanding patches were prepared via diffusion of aqueous solutions of HA and DCF, with different concentrations of DCF, into the wet BNC three-dimensional porous network. The resultant dual polysaccharides-based patches with a nanostructured morphology present thermal stability up to 200 °C, as well as good dynamic mechanical properties, with a storage modulus higher than 1.0 GPa. In addition, the patches are non-cytotoxic to human keratinocytes (HaCaT cells), with a cell viability of almost 100% after 24 h. The in vitro release profile of DCF from the patches was evaluated in simulated saliva, and the data refer to a diffusion- and swelling-controlled drug-release mechanism. The attained results hint at the possibility of using these dual polysaccharides-based oral mucosal patches to target aphthous stomatitis.
Keywords: aphthous stomatitis; bacterial nanocellulose; diclofenac; drug delivery; hyaluronic acid; nanostructured patches.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- Sharma D., Garg R. A comprehensive review on aphthous stomatitis, its types, management and treatment available. J. Dev. Drugs. 2018;7:1000188. doi: 10.4172/2329-6631.1000189. - DOI
-
- Dalessandri D., Zotti F., Laffranchi L., Migliorati M., Isola G., Bonetti S., Visconti L. Treatment of recurrent aphthous stomatitis (RAS; Aphthae; canker sores) with a barrier forming mouth rinse or topical gel formulation containing hyaluronic acid: A retrospective clinical study. BMC Oral Health. 2019;19:153. doi: 10.1186/s12903-019-0850-1. - DOI - PMC - PubMed
Grants and funding
- UIDB/50011/2020 & UIDP/50011/2020/Fundação para a Ciência e a Tecnologia
- UID/AMB/50017/2019/Fundação para a Ciência e a Tecnologia
- CENTRO-01-0145-FEDER-000001/Fundação para a Ciência e a Tecnologia
- SFRH/BD/140230/2018/Fundação para a Ciência e a Tecnologia
- CEECIND/04050/2017/Fundação para a Ciência e a Tecnologia
LinkOut - more resources
Full Text Sources

