Molecular subtypes of oropharyngeal cancer show distinct immune microenvironment related with immune checkpoint blockade response
- PMID: 32235905
- PMCID: PMC7251088
- DOI: 10.1038/s41416-020-0796-8
Molecular subtypes of oropharyngeal cancer show distinct immune microenvironment related with immune checkpoint blockade response
Erratum in
-
Correction: Molecular subtypes of oropharyngeal cancer show distinct immune microenvironment related with immune checkpoint blockade response.Br J Cancer. 2020 Sep;123(6):1041. doi: 10.1038/s41416-020-0944-1. Br J Cancer. 2020. PMID: 32572173 Free PMC article.
Abstract
Background: Oropharyngeal cancer (OPC) exhibits diverse immunological properties; however, their implications for immunotherapy are unknown.
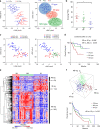
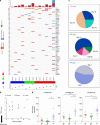
Methods: We analysed 37 surgically resected and nine recurrent or metastatic anti-programmed death-1 (PD-1)/programmed death-ligand 1 (PD-L1)-treated OPC tumours. OPCs were classified into immune-rich (IR), mesenchymal (MS) and xenobiotic (XB) subtypes based on RNA-sequencing data.
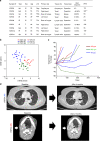
Results: All IR type tumours were human papillomavirus (HPV) positive, most XB types were HPV negative, and MS types showed mixed HPV status. The IR type showed an enriched T cell exhaustion signature with PD-1+ CD8+ T cells and type I macrophages infiltrating the tumour nest on multiplex immunohistochemistry. The MS type showed an exclusion of CD8+ T cells from the tumour nest and high MS and tumour growth factor-β signatures. The XB type showed scant CD8+ T cell infiltration and focal CD73 expression. The IR type was associated with a favourable response signature during anti-PD-1/PD-L1 therapy and showed a high APOBEC mutation signature, whereas the MS and XB types showed resistance signature upregulation. Among anti-PD-1/PD-L1-treated OPC patients, the IR type showed a favourable clinical response (3/4 patients), whereas the XB type showed early progression (3/3 patients).
Conclusion: Our analysis classified OPCs into three subtypes with distinct immune microenvironments that are potentially related to the response to anti-PD-1/PD-L1 therapy.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
- DF-201906-0000001/Daewoong Pharmaceutical Company (Daewoong)
- No. 6-2017-0104/Yonsei University | Yonsei University College of Medicine (YUCM)
- 2017M3A9E802971, 2017M3A9E9072669/National Research Foundation of Korea (NRF)
- HA16C0015020019/Ministry of Health and Welfare (Ministry of Health, Welfare and Family Affairs)
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

