Anti-inflammatory Effect of Curcuma longa and Allium hookeri Co-treatment via NF-κB and COX-2 Pathways
- PMID: 32235914
- PMCID: PMC7109078
- DOI: 10.1038/s41598-020-62749-7
Anti-inflammatory Effect of Curcuma longa and Allium hookeri Co-treatment via NF-κB and COX-2 Pathways
Abstract
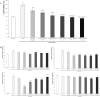
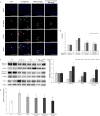
Although inflammation is a host defense mechanism, chronic inflammation mediates several diseases, including cancer, allergy, asthma, and autoimmune diseases, and reportedly, it is associated with a 60% mortality rate. There are several reports on the anti-inflammatory effects of Curcuma longa and Allium hookeri. However, although they can be used as culinary materials and have biological effects, they are not effective anti-inflammatory agents. In this study, we evaluated the synergic effect of C. longa and A. hookeri in order to confirm the possibility of a new anti-inflammatory agent. Based on cell viability and cytokine analyses, the appropriate ratio of C. longa and A. hookeri was confirmed using an air pouch animal model. Then, the anti-inflammatory effect of C. longa and A. hookeri co-treatment was evaluated by measuring the immune cell count and cytokines in the exudate and by comparing the morphological changes and cytokines in inflamed skin samples. Additionally, we evaluated the NF-κB/COX-2 pathway and iNOS levels. The active constituents detected in C. longa were demethoxycurcumin and bisdemethoxycurcumin, and that detected in A. hookeri was methylsulfonylmethane. An in vitro assessment determined the appropriate drug ratio as 3:7. In a carrageenan-induced inflammatory model, co-treatment effectively suppressed inflammatory cytokines, including IFN-γ, IL-1β, IL-6, IL-13, and IL-17, and recovered inflammation-related morphological changes in the skin. The anti-inflammatory effect of the co-treatment was mediated through the NF-κB/COX-2 pathway and iNOS inhibition. We concluded that co-treatment with C. longa and A. hookeri synergistically inhibited inflammation via the NF-κB/COX-2/iNOS pathway.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Pahwa, R., Jialal, I., Chronic inflammation. StatPearls [Internet], https://www.ncbi.nlm.nih.gov/books/NBK493173 (June 4, 2019). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

