Effects of ertugliflozin on renal function over 104 weeks of treatment: a post hoc analysis of two randomised controlled trials
- PMID: 32236732
- PMCID: PMC7228910
- DOI: 10.1007/s00125-020-05133-4
Effects of ertugliflozin on renal function over 104 weeks of treatment: a post hoc analysis of two randomised controlled trials
Abstract
Aims/hypothesis: This study aimed to evaluate the effect of ertugliflozin, a sodium-glucose cotransporter 2 (SGLT2) inhibitor, on eGFR and albuminuria (urine albumin/creatinine ratio [UACR]) vs glimepiride or placebo/glimepiride (non-ertugliflozin) over 104 weeks of treatment in participants with type 2 diabetes mellitus, using pooled data from two randomised controlled, active comparator studies from the eValuation of ERTugliflozin effIcacy and Safety (VERTIS) programme (Clinicaltrials.gov NCT01999218 [VERTIS SU] and NCT02033889 [VERTIS MET]). In the VERTIS SU study, ertugliflozin was evaluated vs glimepiride over 104 weeks. In the VERTIS MET study, ertugliflozin was evaluated vs placebo over 26 weeks; eligible participants were switched from placebo to blinded glimepiride from week 26 to week 104. The glycaemic efficacy of ertugliflozin vs non-ertugliflozin was also assessed in the pooled population.
Methods: Post hoc, exploratory analysis was used to investigate mean changes from baseline in eGFR and UACR over 104 weeks.
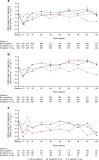
Results: Overall, mean (SD) baseline eGFR was 88.2 (18.8) ml min-1 (1.73 m)-2 and geometric mean (95% CI) of baseline UACR was 1.31 mg/mmol (1.23, 1.38). At week 6, the changes in eGFR from baseline were -2.3, -2.7 and -0.7 ml min-1 (1.73 m)-2 for the ertugliflozin 5 mg, ertugliflozin 15 mg and non-ertugliflozin groups, respectively. Mean eGFR in the ertugliflozin groups increased over time thereafter, while it decreased in the non-ertugliflozin group. Week 104 changes in eGFR from baseline were -0.2, 0.1 and -2.0 ml min-1 (1.73 m)-2 for the ertugliflozin 5 mg, ertugliflozin 15 mg and non-ertugliflozin groups, respectively. Among 415 patients (21.4% of the cohort) with albuminuria at baseline, the ertugliflozin groups had greater reductions in UACR at all measured time points up to week 104. At week 104, the non-ertugliflozin-corrected difference in UACR (95% CI) was -29.5% (-44.8, -9.8; p < 0.01) for ertugliflozin 5 mg and -37.6% (-51.8, -19.2; p < 0.001) for ertugliflozin 15 mg. Least squares mean changes from baseline in HbA1c (mmol/mol [95% CI]) at week 104 were similar between treatment groups: -6.84 (-7.64, -6.03), -7.74 (-8.54, -6.94) and -6.84 (-7.65, -6.03) in the ertugliflozin 5 mg, ertugliflozin 15 mg and non-ertugliflozin groups, respectively. Least squares mean changes from baseline in HbA1c (% [95% CI]) at week 104 were: -0.63 (-0.70, -0.55), -0.71 (-0.78, -0.64) and -0.63 (-0.70, -0.55) in the ertugliflozin 5 mg, ertugliflozin 15 mg and non-ertugliflozin groups, respectively.
Conclusions/interpretation: Ertugliflozin reduced eGFR at week 6, consistent with the known pharmacodynamic effects of SGLT2 inhibitors on renal function. Over 104 weeks, eGFR values returned to baseline and were higher with ertugliflozin compared with non-ertugliflozin treatment, even though changes in HbA1c did not differ between the groups. Ertugliflozin reduced UACR in patients with baseline albuminuria.
Trial registration: clinicaltrials.gov NCT01999218 and NCT02033889.
Keywords: Albuminuria; Diabetic nephropathies; Ertugliflozin; Glomerular filtration rate; Type 2 diabetes mellitus.
Figures

References
-
- Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;377(7):644–657.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

