The impact of pregnancy on the pharmacokinetics of antidepressants: a systematic critical review and meta-analysis
- PMID: 32238008
- PMCID: PMC7323120
- DOI: 10.1080/17425255.2020.1750598
The impact of pregnancy on the pharmacokinetics of antidepressants: a systematic critical review and meta-analysis
Abstract
Introduction: Pregnancy-related physiological changes exert a crucial impact on the pharmacokinetics of antidepressants; however, the current evidence presents inconsistencies. A clearer understanding of pregnancy-related effects on antidepressant disposition may facilitate the development of guidelines for appropriate dose adjustments during the course of pregnancy based on therapeutic drug monitoring.Areas covered: We systematically reviewed studies comparing antidepressant levels in the same individuals during pregnant and non-pregnant states. Using dose-adjusted plasma concentration measurements, we estimated alteration ratios between the 3rd trimester and baseline (before or after pregnancy). Additionally, we performed a meta-analysis for changes in dose-adjusted concentrations to estimate mean differences.Expert opinion: Data for several antidepressants display clear alteration patterns during pregnancy. On the basis of the alteration ratios trimipramine, fluvoxamine, and nortriptyline show a prominent decrease in dose-adjusted levels, especially in the 3rd trimester. Clomipramine, imipramine, citalopram, and paroxetine show smaller decreases in dose-adjusted concentrations in the third trimester. For escitalopram, venlafaxine and fluoxetine, changes are considered negligible. For sertraline, there was a tendency toward increased dose-adjusted concentrations in pregnancy. Available evidence suffers from major limitations and factors affecting pharmacokinetics have been insufficiently addressed. Further research is required to promote knowledge on pregnancy effects on antidepressant pharmacokinetics.
Keywords: Antidepressants; citalopram; fluoxetine; gestation; pharmacokinetics; pregnancy; sertraline; therapeutic drug monitoring; venlafaxine; women’s mental health.
Conflict of interest statement
Declaration of interest
M Paulzen has received speaker’s fee from Neuraxpharma, Langenfeld, Germany. KM Deligiannidis has received research grant support as a site for the clinical trials of brexanolone and zuranolone and serves as a consultant for Sage Therapeutics. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Figures
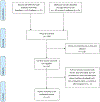


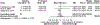
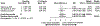
References
-
-
Westin AA, Reimers A, Spigset O. Should pregnant women receive lower or higher medication doses? Tidsskr Nor Laegeforen. 2018. October 30;138(17).
*One of the most comprehensive reviews addressing the need for dose changing for antidepressants during pregnancy.
-
-
-
Deligiannidis KM, Byatt N, Freeman MP. Pharmacotherapy for mood disorders in pregnancy: a review of pharmacokinetic changes and clinical recommendations for therapeutic drug monitoring. J Clin Psychopharmacol. 2014. April;34(2):244–255.
*An early review that reviewed TDM data to provide clinical recommendations for dose selection during pregnancy.
-
-
- Tasnif Y, Morado J, Hebert MF. Pregnancy-related pharmacokinetic changes. Clin Pharmacol Ther. 2016. July;100(1):53–62. - PubMed
-
- Westin AA. The impact of pregnancy on maternal serum concentrations of antiepileptic, antipsychotic and antidepressant drugs. Evidence from therapeutic drug monitoring. Trodheim: Norwegian University of Science and Technology; 2018.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
