A knock-in mouse model for KCNQ2-related epileptic encephalopathy displays spontaneous generalized seizures and cognitive impairment
- PMID: 32239694
- PMCID: PMC7317210
- DOI: 10.1111/epi.16494
A knock-in mouse model for KCNQ2-related epileptic encephalopathy displays spontaneous generalized seizures and cognitive impairment
Abstract
Objective: Early onset epileptic encephalopathy with suppression-burst is one of the most severe epilepsy phenotypes in human patients. A significant proportion of cases have a genetic origin, and the most frequently mutated gene is KCNQ2, encoding Kv7.2, a voltage-dependent potassium channel subunit, leading to so-called KCNQ2-related epileptic encephalopathy (KCNQ2-REE). To study the pathophysiology of KCNQ2-REE in detail and to provide a relevant preclinical model, we generated and described a knock-in mouse model carrying the recurrent p.(Thr274Met) variant.
Methods: We introduced the p.(Thr274Met) variant by homologous recombination in embryonic stem cells, injected into C57Bl/6N blastocysts and implanted in pseudopregnant mice. Mice were then bred with 129Sv Cre-deleter to generate heterozygous mice carrying the p.(Thr274Met), and animals were maintained on the 129Sv genetic background. We studied the development of this new model and performed in vivo electroencephalographic (EEG) recordings, neuroanatomical studies at different time points, and multiple behavioral tests.
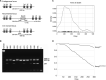
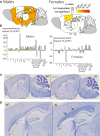
Results: The Kcnq2Thr274Met/+ mice are viable and display generalized spontaneous seizures first observed between postnatal day 20 (P20) and P30. In vivo EEG recordings show that the paroxysmal events observed macroscopically are epileptic seizures. The brain of the Kcnq2Thr274Met/+ animals does not display major structural defects, similar to humans, and their body weight is normal. Kcnq2Thr274Met/+ mice have a reduced life span, with a peak of unexpected death occurring for 25% of the animals by 3 months of age. Epileptic seizures were generally not observed when animals grew older. Behavioral characterization reveals important deficits in spatial learning and memory in adults but no gross abnormality during early neurosensory development.
Significance: Taken together, our results indicate that we have generated a relevant model to study the pathophysiology of KCNQ2-related epileptic encephalopathy and perform preclinical research for that devastating and currently intractable disease.
Keywords: KCNQ2; epileptic encephalopathy; mouse model.
© 2020 The Authors. Epilepsia published by Wiley Periodicals, Inc. on behalf of International League Against Epilepsy.
Conflict of interest statement
None of the authors has any conflict of interest to disclose. We confirm that we have read the Journal's position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
Figures




References
-
- Weckhuysen S, Mandelstam S, Suls A, et al. KCNQ2 encephalopathy: emerging phenotype of a neonatal epileptic encephalopathy. Ann Neurol. 2012;71:15–25. - PubMed
-
- Saitsu H, Kato M, Koide A, et al. Whole exome sequencing identifies KCNQ2 mutations in Ohtahara syndrome. Ann Neurol. 2012;72:298–300. - PubMed
-
- Milh M, Lacoste C, Cacciagli P, et al. Variable clinical expression in patients with mosaicism for KCNQ2 mutations. Am J Med Genet. 2015;167A:2314–8. - PubMed
-
- Morris R. Developments of a water‐maze procedure for studying spatial learning in the rat. J Neurosci Methods. 1984;11:47–60. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

