Iron is a ligand of SecA-like metal-binding domains in vivo
- PMID: 32241912
- PMCID: PMC7247292
- DOI: 10.1074/jbc.RA120.012611
Iron is a ligand of SecA-like metal-binding domains in vivo
Abstract
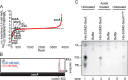
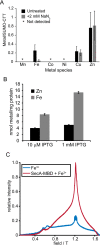
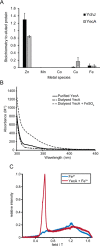
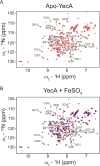
The ATPase SecA is an essential component of the bacterial Sec machinery, which transports proteins across the cytoplasmic membrane. Most SecA proteins contain a long C-terminal tail (CTT). In Escherichia coli, the CTT contains a structurally flexible linker domain and a small metal-binding domain (MBD). The MBD coordinates zinc via a conserved cysteine-containing motif and binds to SecB and ribosomes. In this study, we screened a high-density transposon library for mutants that affect the susceptibility of E. coli to sodium azide, which inhibits SecA-mediated translocation. Results from sequencing this library suggested that mutations removing the CTT make E. coli less susceptible to sodium azide at subinhibitory concentrations. Copurification experiments suggested that the MBD binds to iron and that azide disrupts iron binding. Azide also disrupted binding of SecA to membranes. Two other E. coli proteins that contain SecA-like MBDs, YecA and YchJ, also copurified with iron, and NMR spectroscopy experiments indicated that YecA binds iron via its MBD. Competition experiments and equilibrium binding measurements indicated that the SecA MBD binds preferentially to iron and that a conserved serine is required for this specificity. Finally, structural modeling suggested a plausible model for the octahedral coordination of iron. Taken together, our results suggest that SecA-like MBDs likely bind to iron in vivo.
Keywords: Sec pathway; iron; metal ion-protein interaction; protein secretion; protein structure; protein translocation.
© 2020 Cranford-Smith et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures
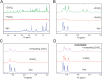
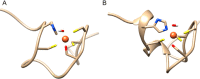
Similar articles
-
The C-terminal tail of the bacterial translocation ATPase SecA modulates its activity.Elife. 2019 Jun 27;8:e48385. doi: 10.7554/eLife.48385. Elife. 2019. PMID: 31246174 Free PMC article.
-
Cellular dynamics of the SecA ATPase at the single molecule level.Sci Rep. 2021 Jan 14;11(1):1433. doi: 10.1038/s41598-021-81081-2. Sci Rep. 2021. PMID: 33446830 Free PMC article.
-
The molecular mechanism of cotranslational membrane protein recognition and targeting by SecA.Nat Struct Mol Biol. 2019 Oct;26(10):919-929. doi: 10.1038/s41594-019-0297-8. Epub 2019 Sep 30. Nat Struct Mol Biol. 2019. PMID: 31570874 Free PMC article.
-
SecA - a multidomain and multitask bacterial export protein.Acta Biochim Pol. 2021 Aug 31;68(3):427-436. doi: 10.18388/abp.2020_5761. Acta Biochim Pol. 2021. PMID: 34463460 Review.
-
SecA-mediated targeting and translocation of secretory proteins.Biochim Biophys Acta. 2014 Aug;1843(8):1466-74. doi: 10.1016/j.bbamcr.2014.02.014. Epub 2014 Feb 25. Biochim Biophys Acta. 2014. PMID: 24583121 Review.
Cited by
-
Continuous Production of Human Epidermal Growth Factor Using Escherichia coli Biofilm.Front Microbiol. 2022 Apr 12;13:855059. doi: 10.3389/fmicb.2022.855059. eCollection 2022. Front Microbiol. 2022. PMID: 35495696 Free PMC article.
-
How Quality Control Systems AID Sec-Dependent Protein Translocation.Front Mol Biosci. 2021 Apr 13;8:669376. doi: 10.3389/fmolb.2021.669376. eCollection 2021. Front Mol Biosci. 2021. PMID: 33928127 Free PMC article. Review.
-
Plastid Molecular Chaperone HSP90C Interacts with the SecA1 Subunit of Sec Translocase for Thylakoid Protein Transport.Plants (Basel). 2024 May 1;13(9):1265. doi: 10.3390/plants13091265. Plants (Basel). 2024. PMID: 38732479 Free PMC article.
-
Pore-Forming Toxins During Bacterial Infection: Molecular Mechanisms and Potential Therapeutic Targets.Drug Des Devel Ther. 2021 Sep 7;15:3773-3781. doi: 10.2147/DDDT.S322393. eCollection 2021. Drug Des Devel Ther. 2021. PMID: 34522083 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
- 099149/Z/12/Z /WT_/Wellcome Trust/United Kingdom
- BB/L019434/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BB/P009840/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- WT_/Wellcome Trust/United Kingdom
- 099185/Z/12/Z/WT_/Wellcome Trust/United Kingdom
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

