Chronology of cellular events related to mitochondrial burnout leading to cell death in Fuchs endothelial corneal dystrophy
- PMID: 32242036
- PMCID: PMC7118119
- DOI: 10.1038/s41598-020-62602-x
Chronology of cellular events related to mitochondrial burnout leading to cell death in Fuchs endothelial corneal dystrophy
Abstract
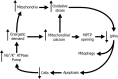
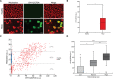
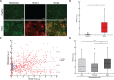
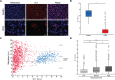
Fuchs endothelial corneal dystrophy (FECD) is a degenerative eye disease characterized by corneal endothelial cell (CEC) death and the formation of guttae, an abnormal thickening of CEC's basement membrane. At the tissue level, an oxidative stress causing mitochondrial damage and CEC death have been described to explain FECD pathogenesis. At the cellular level, our group has previously observed significant variability in the mitochondrial mass of FECD CECs. This led us to hypothesize that mitochondrial mass variability might play a key role in the chronology of events eventually leading to CEC death in FECD. We thus used different fluorescent markers to assess mitochondrial health and functionality as a function of mitochondrial mass in FECD corneal endothelial explants, namely, intra-mitochondrial calcium, mitochondrial membrane potential, oxidation level and apoptosis. This has led us to describe for the first time a sequence of events leading to what we referred to as a mitochondrial burnout, and which goes as follow. FECD CECs initially compensate for endothelial cell losses by incorporating mitochondrial calcium to help generating more ATP, but this leads to increased oxidation. CECs then resist the sustained need for more ATP by increasing their mitochondrial mass, mitochondrial calcium and mitochondrial membrane potential. At this stage, CECs reach their maximum capacity and start to cope with irreversible oxidative damage, which leads to mitochondrial burnout. This burnout is accompanied by a dissipation of the membrane potential and a release of mitochondrial calcium, which in turn leads to cell death by apoptosis.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

